 March
03
March
03
Tags
Why does diabetes make my foot go numb?
Context–what is diabetic neuropathy?
Worldwide, about 1 in 10 adults have diabetes, with that number expected to rise in the coming decades [1]. In this disease, the body becomes unable to properly produce or respond to insulin, leading to high blood sugar levels and abnormal energy processing [2]. Thankfully, many medications, lifestyle interventions (e.g. exercise), and diets have helped millions of individuals manage their diabetes and have an increased quality of life [3]. However, complications of the disease can still arise depending on a multitude of risk factors such as age, time since diagnosis, and how well the diabetes has been managed. The most common complication is damage to the nerves of the peripheral nervous system (Figure 1), which affects about half of all diabetics at some point in their lifetime [4].
Called diabetic neuropathy (DN), this condition is caused primarily by damage to the nerves that carry sensory information from your hands and feet to your brain. While symptoms vary somewhat, symptoms of DN are often categorized into two major subgroups which both affect the hands, legs, and/or feet. Patients in the first subgroup will often report feelings of numbness or tingling. Those who experience the other, painful form of DN called neuropathic pain (30-50% of cases) usually feel a spontaneous (e.g. without necessarily touching anything) burning sensation or severe pain to things that usually wouldn’t cause pain, like light touches [4]. These individuals usually also experience numbness simultaneously, causing them to paradoxically be unable to feel the touch of most things, but still feel pain when touching nothing.
But how exactly does this happen? After all, diabetes is a disease in which the main characteristic is the inability to regulate blood sugar. What does the level of sugar in a person’s blood have to do with their nerves and the way they feel the world around them? Why on Earth would it cause random spontaneous burning sensations in response to everyday actions? Truth be told, the jury is still out. There is no clear silver bullet explanation for DN, but decades of work in this field has led us to a better understanding of what is going on.
Disease in detail–why this matters
On the surface, numbness and tingling may not seem like the most terrible of symptoms (who hasn’t had their leg fall asleep before?). However, this lack of sensation becomes grave when other systemic effects of diabetes come into play. In researching for this article, I spoke to a nurse in Arizona who commented that her team has to keep a careful eye on where her diabetic patients are. They are worried that the patients will go for a walk outside in 120 degree heat barefoot and severely hurt themselves because they can’t feel the second degree burns actively happening on the bottom of their feet. Take into account the often poor healing rates of wounds in diabetics and you have the perfect storm to brew infections. If an infection becomes uncontrollable, the limb will have to be amputated to prevent the infection from spreading. This has led to DN becoming the leading cause of non-traumatic amputations in the world [5], with one occurring every five and a half minutes in the U.S [4]. All this can occur from even the smallest cut on your foot. Now imagine you can’t feel that cut happening, remaining unaware that it may have become infected, and you begin to understand the seriousness of DN. Amputation’s effect on mobility then worsens the other adverse health effects of diabetes and greatly reduces a person’s quality of life.
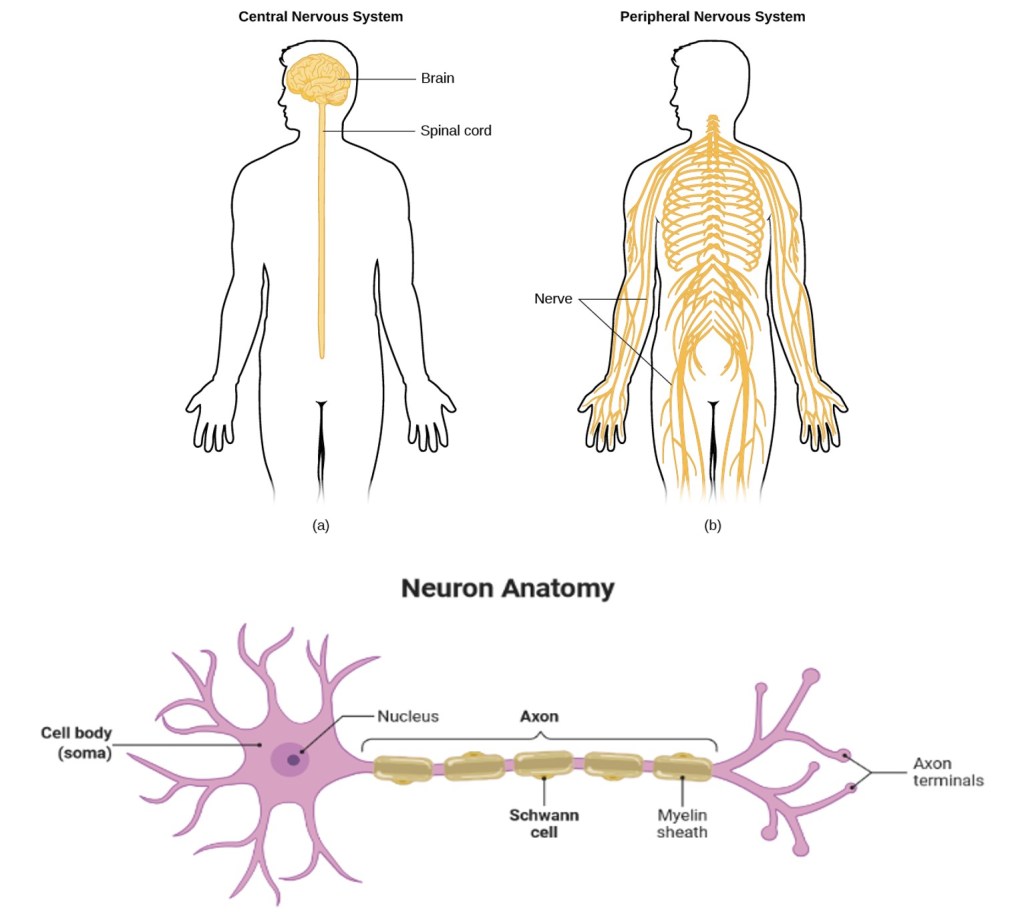
Anatomy of the Peripheral Nervous System (PNS)
To understand DN, we must first understand where it occurs and the players involved. As mentioned previously, DN occurs primarily in the peripheral parts of the body (hands and feet), and is controlled by the peripheral nervous system (PNS), which encompasses all the nerves that carry information to and from the brain, but are located outside of the brain and spinal cord (Figure 1). The communication to and from the brain is carried out by groups of motor and sensory neurons that have their cell bodies near the spine and send projections (called axons) all the way down to their targets (meaning that much of the nerve itself is just bundles of axons). It is within axons that signals are carried from neuron to neuron. The length of axons can be so long that other supporting cells are needed in the nerve to help out the neurons. This is especially true in the case of DN; the two nerves most affected by this condition are also the longest. Arguably the most important of these supporting cells are the Schwann cells, which wrap around the axon and provide vital organization, structure and functional support [6]. Schwann cells come in two types: myelinating and non-myelinating. Myelinating Schwann cells tend to wrap themselves around motor axons to help provide quick conductance of signals.
Sensory axons are largely wrapped by non-myelinating Schwann cells and bundled with other axons of the same type. An easy way to think about this is if you were to imagine the nerve as an electrical wire cable (Figure 2), the myelinating Schwann cells would be the insulation. In addition to the Schwann cells, blood vessels also span the nerve to provide it with oxygen and nutrients. Another main job of these nerve vessels is to ensure that the nerve environment is free from most other molecules found in blood (such as proteins and potential pathogens) so as to create an ideal space for axons to carry signals [7, 8].
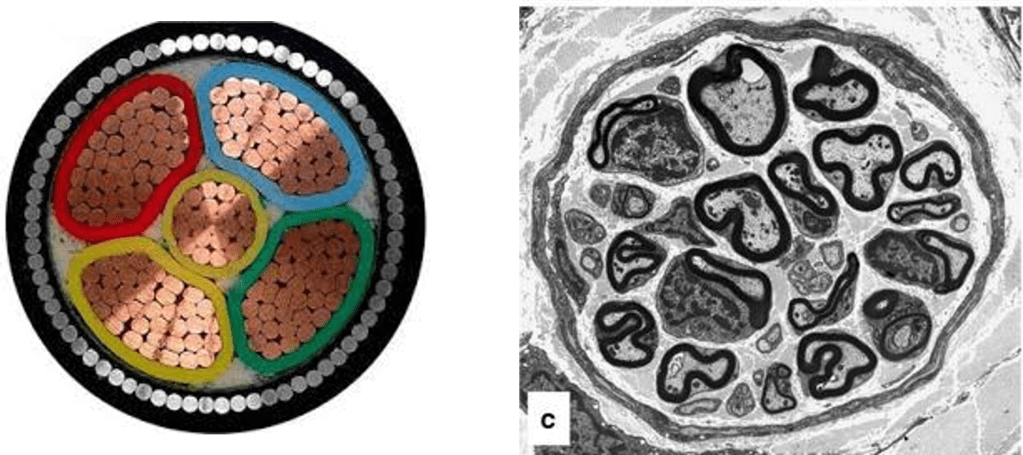
Given the primarily sensory-related symptoms of DN, it is unsurprising that the sensory axons, which carry touch information regarding temperature, pain, and pressure, are most affected by DN. Biopsies from DN patients show that the very tips of the sensory axons seem to degrade first (Figure 3) [4]. This degradation slowly creeps up the axon, while the cell body at the base of the spine remains seemingly healthy [4]. The cell body itself is likely affected by diabetes as well, since diabetes affects the entire human body one way or another. However, it remains unclear whether DN arises due to changes at the cell body or in the axon terminals. Part of the difficulty in understanding DN is figuring out what component of the nerve (axons, Schwann cells, vessels, etc.) is most affected by diabetes and is the primary driver of the symptoms of DN.
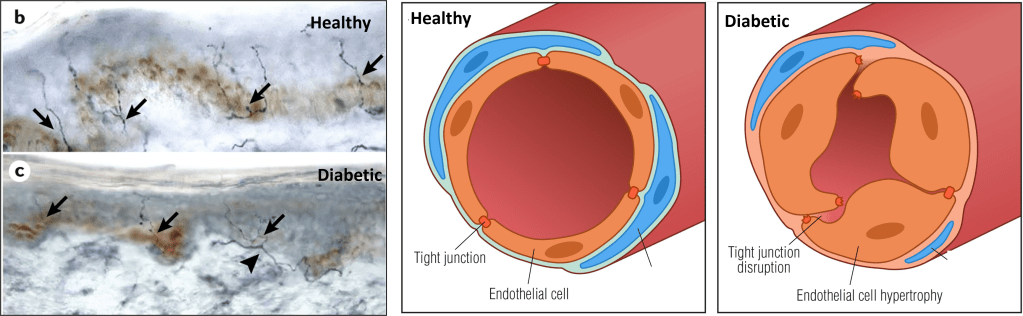
Blood vessels in diabetic nerves are a mess
One way to understand how high blood sugar (among other things) causes the degradation of peripheral nerves is by looking at the peripheral nerve’s blood vessels in diabetic patients and animals. Blood vessels deliver the oxygen and nutrients necessary to keep sensory axons and their Schwann cells functioning. In diabetes, however, endothelial cells, the cells that make up the blood vessels, are swollen and the connections between endothelial cells are lost (Figure 3) [7]. Vessels that are malformed in this way are likely to perform worse at their job. When these connections are lost, proteins and compounds that would normally be found only in the bloodstream enter the nerve [7]. In essence, the nerve’s vessels are leaky. This is worrisome, given the fact that many blood-resident molecules can be toxic to neurons and their axons. In fact, the presence of some of these molecules in the nerve environment have been shown to alter proteins on the axon surface and cause them to fire off a signal much more easily than usual, and thus may contribute to the spontaneous pain sensation symptom [4]. These “misplaced” proteins have been observed in patients before the onset of symptoms, suggesting that vessel leakage may be an initial driver for downstream disease pathways.
However, it’s still debated why the nerve’s vessels behave in this way in diabetes. It could be an increase in inflammatory signals that the whole body experiences during a diabetic state that then causes changes in how endothelial cells operate. Alternatively, it could be the simple consequence of the endothelial cells failing to adapt to the higher levels of circulating glucose found in the blood of diabetic patients.
Metabolic mechanisms
As intriguing as the above vascular hypothesis is, a simpler one may be the answer. In a diabetic state, there are elevated levels of glucose and free fatty acids, which cells normally convert to energy. As such, an overload of these compounds can overwhelm the system. This causes the cells to use secondary reactions to process all of the excess glucose and fatty acids [4]. These reactions, however, are intended as a back up and don’t operate without consequences. In diabetes, the use of these “back up” reactions results in an uptick in inflammatory signals and damaging molecules that harm mitochondria and DNA [4]. This dangerous cascade happens in many cells in the body, but in the case of DN, Schwann cells and the axons they wrap are particularly susceptible. Sensory axons require a lot of energy to send their signals effectively because of their lack of myelination, which may explain why they are affected more than myelinated motor axons. Any bit of dysfunction in the way they or the Schwann cells process energy will affect their ability to carry sensory information up to the brain.
Furthermore, when high levels of glucose are present in the blood, they often react with other molecules in the blood (hemoglobin, cholesterol, etc.) and entirely change their function. Glucose-modified proteins can increase inflammatory signals and mitochondrial dysfunction by acting on a receptor specific to these types of proteins [9]. These can also cause damage to the membranes that line each cell, causing them to lose some structural integrity and hindering communication with neighboring cells. Glucose-modified proteins have also been found to be able to cross into the internal nerve space much more easily than non-modified proteins, where they can directly damage Schwann cells and sensory axons [10]. In fact, injection of glucose-modified proteins over a long period of time into healthy, non-diabetic rats is enough to give those rats neuropathy, suggesting that they are extremely damaging to the nerves [11].
Treatment options
At the moment, there are no disease-modifying medications available to treat DN in the U.S. According to many of the leading experts, well-managed diabetes is currently the best option to slow down the progression of symptoms. This includes controlled blood sugar and increased exercise, with some studies suggesting that some degree of nerve repair actually occurs after exercise regimens [12].
For those that experience the painful type of neuropathy, the American Academy of Neurology has recently recommended the use of anticonvulsants and a type of antidepressants called serotonin-norepinephrine reuptake inhibitors (SNRIs) [13]. At first glance, these may seem like medication to give to someone with psychiatric disease, and not to those experiencing pain in their feet. However, in neuropathy, these drugs serve to interrupt or attenuate the excess pain signaling coming from sensory axons [4]. For example, SNRIs operate by acting on an internal circuit within the spinal cord that uses serotonin and norepinephrine to “quiet” the pain signal coming from sensory axons by increasing their active time in the spinal cord.
Conclusion
The complexity of the peripheral nervous system and diabetes itself makes diabetic neuropathy a difficult problem to unravel. However, the quality of life of many depends on the progress scientists and clinicians are making on this condition. For the nearly half a billion people who suffer with diabetes worldwide (81% of which live in low or middle-income countries [1]), DN can be a debilitating condition that takes away their ability to work and enjoy life to the fullest extent, while dramatically increasing the risk for a life cut short. In all likelihood, each of the hypotheses I outlined today (plus ones I didn’t get to or ones yet to be thought of) are all playing some role in the progression of this disease. Thus, new tools to study this intricacy and see the whole picture of DN are being employed in labs across the world in the hopes of giving people back the feeling of their toes.
References
2. https://www.cdc.gov/diabetes/basics/diabetes.html
3. https://www.mayoclinic.org/diseases-conditions/diabetes/diagnosis-treatment/drc-20371451
4. EL, Feldman. et al. Diabetic neuropathy. Nat. Rev. Dis. Prim. 5, 42 (2019).
5. Diagnosis and management of type 2 diabetes (HEARTS-D). Geneva]: World Health Organization; 2020 (WHO/UCN/NCD/20.1). Licence: CC BY-NC-SA 3.0 IGO.
6. Balakrishnan, A. et al. Insights Into the Role and Potential of Schwann Cells for Peripheral Nerve Repair From Studies of Development and Injury. Front. Mol. Neurosci. 13, 270 (2021).
7. Richner, M. et al. Functional and Structural Changes of the Blood-Nerve-Barrier in Diabetic Neuropathy. Front. Neurosci. 12, 1038 (2019).
8. Mizisin, A. P. & Weerasuriya, A. Homeostatic regulation of the endoneurial microenvironment during development, aging and in response to trauma, disease and toxic insult. Acta Neuropathol. 121, 291 (2011).
9. Goldin, A., Beckman, J. A., Schmidt, A. M. & Creager, M. A. Advanced glycation end products: Sparking the development of diabetic vascular injury. Circulation 114, 597–605 (2006).
10. Poduslo, J. F. & Curran, G. L. Increased permeability across the blood-nerve barrier of albumin glycated in vitro and in vivo from patients with diabetic polyneuropathy. Proc. Natl. Acad. Sci. U. S. A. 89, 2218 (1992).
11. Nishizawa, Y. et al. Neuropathy induced by exogenously administered advanced glycation end‐products in rats. J. Diabetes Investig. 1, 40 (2010).
12. Singleton, J. R. et al. Exercise increases cutaneous nerve density in diabetic patients without neuropathy. Ann. Clin. Transl. Neurol. 1, 844–849 (2014).Price, R. et al. Oral and Topical Treatment of Painful Diabetic Polyneuropathy: Practice Guideline Update Summary. Neurology 98, 31–43 (2022).
13. Price, R. et al. Oral and Topical Treatment of Painful Diabetic Polyneuropathy: Practice Guideline Update Summary. Neurology 98, 31–43 (2022).

Getting diabetic neuropathy is like having ruined nerves or sitting all the time. Sugar will add up to wherever there is a stopping point like hands & feet. Exercise is most of what you need when you have everything in your body working right. I will tell you that to get rid of any & all sugars out of your systems & hands & feet is using cinnamon a lot. That sugar level reducing spice can actually make all that overloading of sugar get straightened out using it instead. Cinnamon will really make it happen!