 February
28
February
28
Tags
The Scientific Adventures of Ben Barres Part II: Superstar of the Star Cells
This post is the second half of a two-part series on the work of Dr. Ben Barres. Did you miss the first half of our adventure? No problem. Click here to start from the beginning!
Welcome back! Last week the adventure through Ben Barres’ lab’s discoveries wound through their exciting advances in cell culture, their generation of hugely important genomic datasets, and some of their work on oligodendrocytes and endothelial cells. Today the star of the show will be the star cells.
Astrocytes: Stars of Synaptic Development
Particularly in more recent years, Ben’s lab is widely known for its seminal work on astrocytes. The word astrocyte comes from the Greek word for “star cell,” and one needs only to see a photo of an astrocyte to understand why this is an apt name. Astrocytes extend several long, thin processes from their cell bodies, wrapping them around blood vessels as well as around synapses, the connection points between two neurons where all the magic happens. It was previously thought that astrocytes had a mostly passive role at synapses, but the field could not have been more wrong.

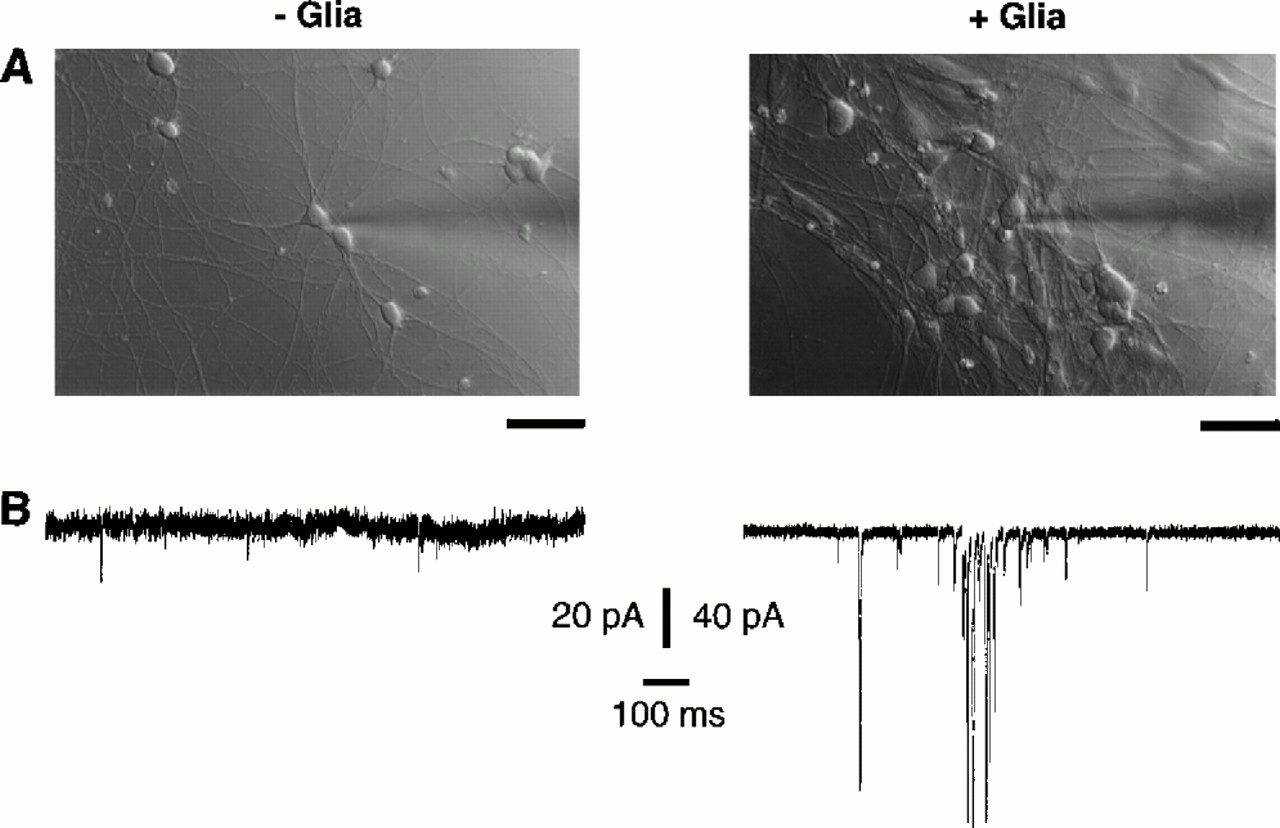
A series of findings from Ben’s lab promptly turned that idea upside-down. As discussed in Part I last week, Ben’s lab had figured out that CNS neurons could survive and extend lots of axons and dendrites in culture. However, despite looking like mature neurons, they didn’t act like mature neurons in that they did not exhibit much spontaneous activity. Normally, neurons spontaneously release signaling chemicals called neurotransmitters every so often, even when not involved in a particular task or thought.
Frank Pfrieger figured out that when he added astrocytes to the culture, the neurons’ spontaneous activity was five times stronger and seventy times more frequent, and the neurons released twelve times as much neurotransmitter [1]! Undoubtedly, astrocytes were playing an important role in neural function. Erik Ullian later realized that adding astrocytes to a neural culture could increase the number of synapses by a factor of seven. Interestingly, Erik also found that the formation of synapses in the developing brain occurred around the same time that astrocytes were generated [2]. Could it be that neural synapses—the connections that are the basis for all neural function—wouldn’t exist without the “passive” star-shaped cell?! Indeed.
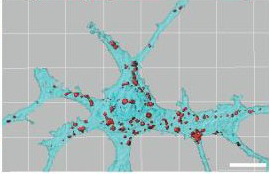
Karen Christopherson figured out that astrocytes were instructing neurons to form connections with one another by releasing signals called thrombospondins [3]. Thrombospondins from astrocytes attach to special receptors on neurons (receptors identified by Cagla Eroglu), and induce the neurons to form mature, structurally sound synapses [4]. But strangely, these synapses were silent; although the neurons were in place to talk to one another, they weren’t able to do so. What extra factor was needed to give the neurons the functional ability to communicate?
Nicola Allen solved this mystery. She found that while postsynaptic neurons (signal recipients) made the receptors necessary to receive neurotransmitter signals from their presynaptic partners (signal senders), in the absence of astrocytes, these receptors stayed on the inside of the cell. Without access to the outside of the cell, these receptors are essentially useless. Molecules released from astrocytes—which Nicola identified as Glypicans 4 and 6—caused the neurons to immediately shuttle these neurotransmitter receptors to their surfaces, allowing them to start receiving messages from their synaptic partners [5]. (For more on this story and the work that Nicola has continued in her own lab, check out Elena’s post on astrocytes!)

Hungry for Synapses
Astrocytes are not only important in creating synapses, but also in destroying them. From mice to men, mammals form too many synapses during development. For healthy development, many of these synapses need to be pruned so that the rest can function more precisely. Many believe that a lack of proper pruning underlies developmental disorders such as autism spectrum disorders. Beth Stevens helped to identify how neurons tag themselves for destruction during this pruning process [6]. They spit out a molecule called C1q, and are then destroyed. How exactly? Microglia, the immune cells of the central nervous system accustomed to eating debris, seemed likely candidates to be the ones eating these synapses.
While Beth found that microglia do indeed eat many of these extra synapses during development [6], Won-Suk Chung discovered that, surprisingly, our “passive” astrocytes also help to prune the unnecessary synapses. In fact, in the presence of astrocytes, developing neurons make and spit out more C1q than they do on their own. And while microglia lose their ability to eat synapses in the healthy adult, astrocytes retain their gobbling ability past development and continue to be able to do this at a low level in adult brain, preferentially pruning synapses that are less active [7]. This gives the astrocytes the power to actively sculpt neural networks, keeping them clean and working properly.
Astrocytes: Good or Evil?
Astrocytes are clearly essential for the development and continual working of a healthy brain. However, the Barres lab recently uncovered evidence that astrocytes sometimes cross over to the dark side, murdering the neurons that they normally help to nurture.
It had been known that astrocytes become “reactive” when something goes wrong in the brain, undergoing a change in shape and rushing to the scene of the crime. But it was somewhat unclear whether they were rushing to keep the peace or contributing to the ruckus. Jennifer Zamanian was surprised to find that reactive astrocytes come in very distinct flavors. In a model of stroke, astrocytes released a bunch of molecules known to support neuronal health. In a model of severe infection, however, astrocytes were creating molecules used to tag neurons for destruction [8]. This stark dichotomy led to the idea that reactive astrocytes can be broadly categorized into bad (“A1”) and good (“A2”) astrocytes.
Shane Liddelow identified that three special molecules released from microglia were sufficient to turn resting astrocytes into evil A1 astrocytes that then lost many of their functions that lend support to neurons and synaptic connections [9]. A1 astrocytes also start releasing a neurotoxin that kills any injured neurons in the area while sparing the strong, healthy ones. (No one yet knows what this neurotoxin actually is; Ben’s graduate student Kevin Guttenplan is currently working to uncover its identity.)
Might A1 astrocytes play an important role in human disease? When he looked at postmortem tissue from humans who had died from neurodegenerative diseases, Shane found these A1 astrocytes were abundant across the board: brains from patients with Alzheimer’s, Parkinson’s, and Huntington’s diseases, amyloid lateral sclerosis (ALS), and multiple sclerosis (MS) all had evidence of A1 astrocytes. Thus, evil astrocytes might be killing struggling neurons in these diseases, hastening the devastating symptoms that arise from widespread neuronal death.

The Two-Hit Hypothesis
Why is age such a big risk factor for neurodegenerative disease? Ali Stephan created a new tool to examine which cells in the brain are tagged with C1q, the “eat me” signal found on synapses that need to be pruned during development. Shockingly, Ali found lots of C1q on synapses in the aging mouse brain, earliest in the hippocampus (the memory-related brain area particularly vulnerable to Alzheimer’s disease) and then spreading elsewhere [10].
This led Ben Barres to a “two-hit hypothesis”: old, perhaps underutilized synapses in the aging brain are tagged with C1q. This age-related tagging is the first hit. A second hit—an acute or chronic neural injury, or neural inflammation—might cause the generation of A1 astrocytes. The C1q tag makes synapses vulnerable to death by the mysterious neurotoxin released by A1 astrocytes. When many neurons begin succumbing to the neurotoxin, we begin to see the clinical manifestations of neurodegenerative disorders.

If we could get rid of the C1q tag on aging synapses, could we save them from death during the second hit by A1 astrocytes? To be actively involved in the pursuit of this question, Ben co-founded a biotech company, Annexon Biosciences, with Arnon Rosenthal in 2011. The drug that they made to target C1q has been successful in several mouse models of neurological disease, including neuromyelitis optica, Guillian-Barre Syndrome, spinal muscular atrophy, and Alzheimer’s disease. Its clinical safety is now being tested in human patients! If successful, it has the potential to be effective in slowing not just one disease, but many.
An End, but Not Really
Ben Barres passed away in December, 2017 from pancreatic cancer. Scientists across the world mourned the death of a brilliant mind, dedicated mentor, and ruthless advocate for equity and inclusivity in science.
However, Ben’s legacy lives on. An incredible number of his trainees are now leading their own labs, and each day they continue to explore the questions that they first started to ask under Ben’s guidance.
Ben, of course, gives them all the credit: “In an academic lab at a top university like Stanford, the principal investigator (PI) is not the one doing the experiments and not even the one having most of the ideas (if I am lucky I get to make an occasional suggestion that is not instantly thrown under the bus). I can honestly say that the vast majority of my graduate students and postdoctoral fellows have been far more talented than I ever was.”
I’d like to give Ben more credit than he was willing to give himself. Thank you, Ben, for inspiring and guiding an incredible number of scientific discoveries that pushed neuroscience forward in leaps and bounds. Thank you for shining a light on the importance of glial cells, the undervalued members of the CNS that create and support the neural synapses underlying our every move and thought. Thank you for teaching each of your trainees the importance of asking big questions, and for giving them an example of stellar mentorship. Your legacy lives on through them and through all those you inspired during your amazing adventure of scientific discovery.

References
1. Pfrieger FW, Barres BA. Synaptic efficacy enhanced by glial cells in vitro. Science. 1997 Sep 12;277(5332):1684-7. PubMed PMID: 9287225.
2. Ullian EM, Sapperstein SK, Christopherson KS, Barres BA. Control of synapse number by glia. Science. 2001 Jan 26;291(5504):657-61. PubMed PMID: 11158678.
3. Christopherson KS, Ullian EM, Stokes CC, Mullowney CE, Hell JW, Agah A, Lawler J, Mosher DF, Bornstein P, Barres BA. Thrombospondins are astrocyte-secreted proteins that promote CNS synaptogenesis. Cell. 2005 Feb 11;120(3):421-33. PubMed PMID: 15707899.
4. Eroglu C, Allen NJ, Susman MW, O’Rourke NA, Park CY, Ozkan E, Chakraborty C,Mulinyawe SB, Annis DS, Huberman AD, Green EM, Lawler J, Dolmetsch R, Garcia KC, Smith SJ, Luo ZD, Rosenthal A, Mosher DF, Barres BA. Gabapentin receptor alpha2delta-1 is a neuronal thrombospondin receptor responsible for excitatory CNS synaptogenesis. Cell. 2009 Oct 16;139(2):380-92. doi:10.1016/j.cell.2009.09.025. Epub 2009 Oct 8. PubMed PMID: 19818485; PubMed Central PMCID: PMC2791798.
5. Allen NJ, Bennett ML, Foo LC, Wang GX, Chakraborty C, Smith SJ, Barres BA. Astrocyte glypicans 4 and 6 promote formation of excitatory synapses via GluA1 AMPA receptors. Nature. 2012 May 27;486(7403):410-4. doi: 10.1038/nature11059. PubMed PMID: 22722203; PubMed Central PMCID: PMC3383085.
6. Stevens B, Allen NJ, Vazquez LE, Howell GR, Christopherson KS, Nouri N, Micheva KD, Mehalow AK, Huberman AD, Stafford B, Sher A, Litke AM, Lambris JD, Smith SJ, John SW, Barres BA. The classical complement cascade mediates CNS synapse elimination. Cell. 2007 Dec 14;131(6):1164-78. PubMed PMID: 18083105.
7. Chung WS, Clarke LE, Wang GX, Stafford BK, Sher A, Chakraborty C, Joung J, Foo LC, Thompson A, Chen C, Smith SJ, Barres BA. Astrocytes mediate synapse elimination through MEGF10 and MERTK pathways. Nature. 2013 Dec 19;504(7480):394-400. doi: 10.1038/nature12776. Epub 2013 Nov 24. PubMed PMID:24270812; PubMed Central PMCID: PMC3969024.
8. Zamanian JL, Xu L, Foo LC, Nouri N, Zhou L, Giffard RG, Barres BA. Genomic analysis of reactive astrogliosis. J Neurosci. 2012 May 2;32(18):6391-410. dos: 10.1523/JNEUROSCI.6221-11.2012. PubMed PMID: 22553043; PubMed Central PMCID:PMC3480225.
9. Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Münch AE, Chung WS, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Buckwalter MS, Rowitch DH, Dawson VL, Dawson TM, Stevens B, Barres BA. Neurotoxic reactive astrocytes are induced by activated microglia.Nature. 2017 Jan 26;541(7638):481-487. doi: 10.1038/nature21029. Epub 2017 Jan
18. PubMed PMID: 28099414; PubMed Central PMCID: PMC5404890.
10. Stephan AH, Madison DV, Mateos JM, Fraser DA, Lovelett EA, Coutellier L, Kim L, Tsai HH, Huang EJ, Rowitch DH, Berns DS, Tenner AJ, Shamloo M, Barres BA. A dramatic increase of C1q protein in the CNS during normal aging. J Neurosci. 2013 Aug 14;33(33):13460-74. doi: 10.1523/JNEUROSCI.1333-13.2013. PubMed PMID:23946404; PubMed Central PMCID: PMC3742932.
11. Bushong EA, Martone ME, Ellisman MH. Maturation of astrocyte morphology and the establishment of astrocyte domains during postnatal hippocampal development. Int J Dev Neurosci. 2004 Apr;22(2):73-86. PubMed PMID: 15036382.
12. Liddelow SA, Barres BA. Reactive Astrocytes: Production, Function, and Therapeutic Potential. Immunity. 2017 Jun 20;46(6):957-967. doi:
10.1016/j.immuni.2017.06.006. Review. PubMed PMID: 28636962.

You must be logged in to post a comment.