 March
07
March
07
Tags
The “miracle” of eye movements: EMDR and PTSD
“Hold that image in your mind,” I say, “and follow my finger”. I proceed to wave my finger in front of your face, back and forth, back and forth, back and forth…
Believe it or not, I’m not performing hypnosis in this hypothetical scenario. Instead, I’m describing one of the most successful behavioral treatments for post-traumatic stress disorder (PTSD): “eye movement desensitization and reprocessing”, or EMDR. This treatment differs from other forms of psychotherapy in that it involves alternating sensory stimulation – usually the back-and-forth movement of the clinician’s finger – that the patient is instructed to attend to while holding in mind whatever thought or memory is causing them distress. This is then repeated while the patient holds in mind a set of positive thoughts that they are striving for [1].
While this psychotherapeutic approach has been applied in the clinic with a surprising amount of success for nearly three decades, clinicians have been at a loss as to why it works. After all, there’s no obvious reason to think that alternating eye movements should have any impact on one’s fear and anxiety, let alone a long-lasting ameliorative effect. So how does EMDR work, and how does it compare to other treatment strategies for PTSD? Let’s first step back and take a look at the origin and applications of EMDR.

A visionary new treatment for PTSD?
The story goes that psychologist Francine Shapiro first dreamed up the idea for EMDR during a walk in the park when she just happened to notice that moving her eyes helped with some of her own negative thoughts [2]. After that, she worked on incorporating eye movements into a standardized clinical practice and published her first study in 1989, showing that PSTD patients that underwent a single session of EMDR, as opposed to a “placebo” treatment condition in which patients similarly described their traumatic experience without making alternating eye movements, had better dramatically improved outcomes. Many of these patients’ presenting problems (nightmares, flashbacks, etc.) were eliminated entirely, and importantly, these improvements persisted at least three months post-treatment [2].
Since then, there have been countless follow-up tests to further evaluate the efficacy of EMDR treatment. For instance, a 2014 meta-analysis of many trials of EMDR therapy that were conducted in the prior two decades concluded that EMDR was effective in reducing PTSD symptoms as well as depression, anxiety, and subjective distress in PTSD patients [3].
While EMDR is a generally accepted psychotherapeutic approach to PTSD treatment (it even earned an A rating from the Agency for Health Care Policy and Treatment for PTSD treatment in adults [4]), it is also highly controversial. Many are skeptical whether its core, defining feature – eye movement desensitization – is, in fact, a critical component of the therapy or merely an unnecessary addition to any other form of cognitive behavioral therapy [5]. While some studies have shown that EMDR is more effective than other cognitive behavioral therapy [e.g., 6], even more say that EMDR is effective but not any more so than other therapies [1, 7]. Moreover, even if there is something uniquely beneficial to making eye movements back and forth, whether the benefit is actually unique to eye movements per se or generalizable to any sort of alternating side-to-side sensory stimulation – such as hearing tones presented alternately to each ear through headphones or alternating right and left finger taps – is also in question [8].
Most of these questions arise because we simply don’t know how or why EMDR works. While some hypotheses have been put forward – synchronizing the brain’s two hemispheres, interfering with working memory processes, engaging similar processes to REM sleep – they remain largely speculative and have not obtained critical consensus. In the absence of any understanding of the neurobiological mechanisms underlying EMDR, it’s perhaps unsurprising that it remains highly controversial and is dismissed by some as pseudoscience. If we could better understand what is happening in the brain during EMDR, we could address some of these controversies and perhaps even develop new treatments – such as drugs that target EMDR’s underlying biological mechanisms directly without relying on behavioral interventions. Remarkably, brand-new research has identified just such a mechanism.
A putative circuit underlying EMDR

A group of neuroscience researchers in South Korea recently identified a few critical brain areas that are recruited together specifically by alternating bilateral stimulation (ABS) in such a way that reduces fear [9]. But to be able to study how these brain areas are engaged by ABS at the cellular level (something that is not currently possible in humans), they had to adapt an ABS protocol similar to that used for EMDR to something that would work with mice. Anyone who has ever worked with mice will tell you that moving your finger back and forth in front of their face will accomplish nothing (except possibly getting you bitten). So as a substitute for a therapist waving their finger, the researchers designed a cylindrical chamber with a line of LEDs that were turned on and off sequentially and in alternating directions, creating an effect of light moving back and forth.
Of course, another challenge of using mice to study PTSD treatments is that mice aren’t diagnosed with PTSD. However, there are well-established methods for inducing long-lasting fear of a particular stimulus (e.g., a tone) by pairing it with a traumatic stimulus (e.g., a foot shock). After this process of fear conditioning, mice will freeze with fear whenever they hear that tone because they have learned to associate that tone with getting shocked. Mice will then be put through an “extinction” phase, during which the tone is repeatedly presented without a foot shock (see below figure). Importantly, while mice do begin to freeze less as they hear the tone more times without getting shocked (i.e., their fear is getting “extinguished”), they continue to freeze more than mice who never learned a tone-shock association, even when they are put in an entirely different context from where they first learned the tone-shock association, and even after many days have passed. This might be likened to some veterans experiencing PTSD back in their home environment and long after their traumatic experience.
To test the efficacy of ABS for reducing fear symptoms, the researchers fear-conditioned their mice, and then a day later conducted extinction trials (tones presented without footshock) in a new context with or without ABS. In this way, extinction trials with ABS are similar to a clinical psychologist prompting you to think about the thought or memory that is causing you distress (tone) while waving their finger back and forth (ABS). Sure enough, the mice that got ABS with tone presentations froze much less frequently than the mice that heard the tone without ABS, suggesting that their fear was more effectively extinguished with ABS. Interestingly, neither continuously lit nor flashing LEDs improved fear extinction, so there must be something unique about alternating visual stimulation compared to other types of visual stimulation that makes it especially effective for reducing fear. Finally, to test whether their ABS protocol had long-lasting beneficial effects for fear reduction as has been demonstrated with EMDR for PTSD symptoms, the researchers tested the mice’s freezing behavior in response to the same tone one week later. Even at this later timepoint, the mice that had received ABS during extinction froze less than half as often as the mice who didn’t get ABS, demonstrating that ABS is doing something that has long-lasting changes in the brain to make the mice less fearful (you can see for yourself in this video published by the researchers).
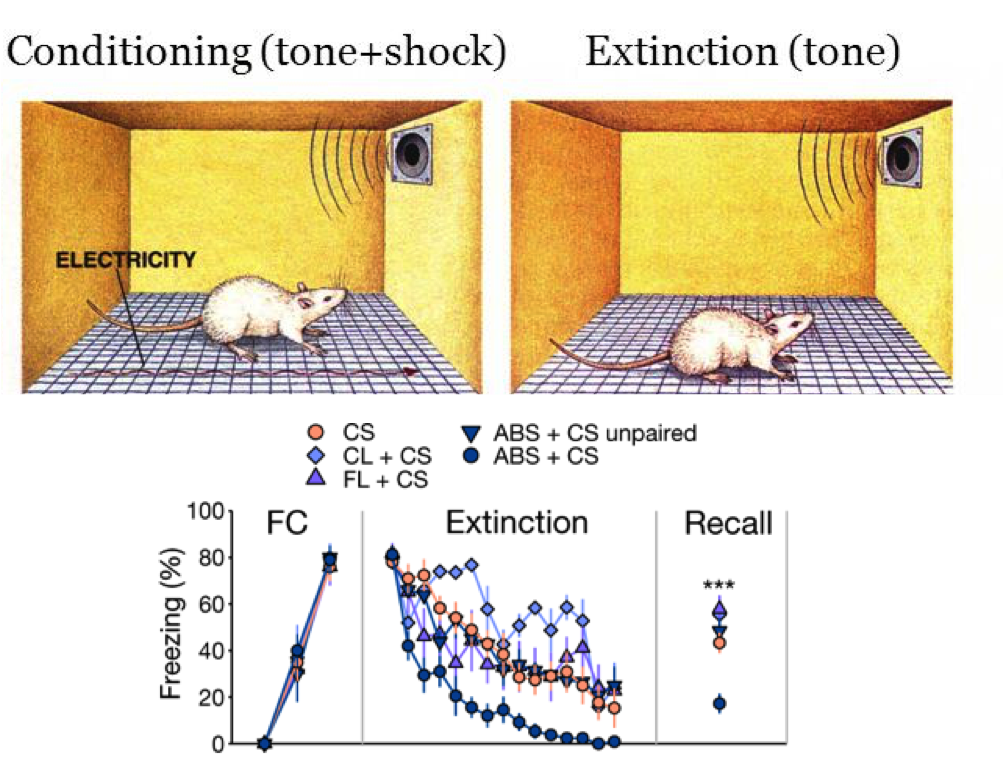
What are these brain changes? The researchers found that both the superior colliculus and the mediodorsal nucleus of the thalamus (MD) were critically important for ABS’s effect of attenuating fear. First, in the superior colliculus – a structure which resides in the back of the brain just above the brainstem and has been shown to be important for visual attention and controlling eye movements – more neurons became excited by the tone paired with ABS compared to the tone alone. Consequently, MD – which receives direct input from the superior colliculus – became activated to a greater degree by the ABS-tone combination. To further demonstrate the importance of this pathway from superior colliculus to MD during ABS, the researchers showed that stimulating this pathway during extinction trials without ABS had the same effect of reducing fear expression, whereas inhibiting that pathway during extinction with ABS counteracted ABS’s positive effects. All in all, the superior colliculus-MD pathway seems likely to be a critical component for ABS’s fear attenuating effects.
But how does excitation of MD translate into reduced fear? MD in turn sends information to the amygdala – the “fear center” of the brain. Many neurons in the amygdala are more active when the animal experiences fear, so you might expect that ABS somehow reduces the activity of these “fear” cells. MD makes that happen; the researchers showed that when MD gets more excited by the superior colliculus during ABS, it increases the amount of inhibition within the amygdala, thus reducing the activity of fear-encoding cells. Importantly, that increased inhibition persists at least a week after fear extinction, meaning that these long-lasting changes in the amygdala could be the key neurological mechanism underlying the long-term positive effects of EMDR.
This is significant because we previously had no idea what might be happening in the brain with EMDR. But now, we have a potential mechanism: alternating bilateral stimulation engages the superior colliculus, which sends greater excitation to MD which in turn causes fear-encoding cells in the amygdala to be inhibited. Having a putative biological mechanism will likely change how many scientists and clinicians looks at EMDR treatment. For instance, this research suggests that the eye movements component of EMDR is, in fact, critical for the treatment’s efficacy because it specifically engages the superior colliculus, whereas other types of visual stimulation do not do so as effectively [9].
Of course, many questions remain. Does EMDR in humans engage the same circuit as alternating bilateral stimulation in mice? Are eye movements necessary for engaging the superior colliculus, or might other types of cognitive behavioral therapies that don’t involve eye movements recruit the same circuit (or even different circuits that produce the same result)? Similarly, are other forms of alternating bilateral stimulation like auditory or tactile equally effective, as has been suggested in humans? There is much more work to be done, but the finding that alternating visual stimulation engages a brain circuit that reduces fear is surprising and compelling. The brain works in mysterious ways, but as they become a little less mysterious, we may be able to improve existing treatments and develop new ones for devastating psychiatric conditions like PTSD.
References:
- Foa, E. B., Keane, T. M., Friedman, M. J., & Cohen, J. A. (2008). Effective Treatments for PTSD, Second Edition: Practice Guidelines from the International Society for Traumatic Stress Studies. Guilford Publications. Retrieved from https://books.google.com/books?id=MFyEg007YEIC
- Shapiro, F. J Trauma Stress (1989) 2: 199. https://doi.org/10.1007/BF00974159
- Chen, Y.-R., Hung, K.-W., Tsai, J.-C., Chu, H., Chung, M.-H., Chen, S.-R., … Chou, K.-R. (2014). Efficacy of Eye-Movement Desensitization and Reprocessing for Patients with Posttraumatic-Stress Disorder: A Meta-Analysis of Randomized Controlled Trials. PLOS ONE, 9(8), e103676. https://doi.org/10.1371/journal.pone.0103676
- https://www.istss.org/ISTSS_Main/media/Documents/ISTSS_g81.pdf
- Davidson, P.R., & Parker, K.C. (2001). Eye movement desensitization and reprocessing (EMDR): a meta-analysis. J Consult Clin Psychol 69: 305–316.
- Chen,L.,Zhang,G.,Hu,M.&Liang,X.Eyemovementdesensitizationand reprocessing versus cognitive-behavioral therapy for adult posttraumatic stress disorder: systematic review and meta-analysis. J. Nerv. Ment. Dis. 203, 443–451 (2015).
- Seidler, G., & Wagner, F. (2006). Comparing the efficacy of EMDR and trauma-focused cognitive-behavioral therapy in the treatment of PTSD: A meta-analytic study. Psychological Medicine, 36(11), 1515-1522. doi:10.1017/S0033291706007963
- Servan-Schreiber, D., Schooler, J., Dew, M.A., Carter, C., & Bartone, P. (2006). Eye Movement Desensitization and Reprocessing for Posttraumatic Stress Disorder: A Pilot Blinded, Randomized Study of Stimulation Type. Psychother Psychosom;75:290-297. doi: 10.1159/000093950
- Baek, J., Lee, S., Cho, T., Kim, S.-W., Kim, M., Yoon, Y., … Shin, H.-S. (2019). Neural circuits underlying a psychotherapeutic regimen for fear disorders. Nature, 566(7744), 339–343. https://doi.org/10.1038/s41586-019-0931-y
Images:
- Featured gif: https://i.gifer.com/HCET.gif
- EMDR gif: https://gfycat.com/scratchydarlinganophelesmosquito
- Fear conditioning schematic: https://images.slideplayer.com/13/3781890/slides/slide_21.jpg

Pingback: Brainspotting: Are the eyes the window to the soul? | NeuWrite San Diego