 March
21
March
21
Tags
Ketamine: A New Hope
You may know ketamine as “Special K,” a party drug with a niche in the Burning Man and rave scenes. It’s known for its dissociative effects–its ability to make its users feel as if they are floating, detached from their bodies and surroundings. It’s a quick trip; the main dissociative effects of ketamine only last up to 25 minutes. This short-lived nature can unfortunately lead to binging, increased tolerance, dependence, and withdrawal symptoms. At high doses, ketamine can lead to complete dissociation and blackout, aka falling into a “K-hole.”
You also may have seen ketamine in the news headlines recently, albeit in a very different context. On March 5th, 2019, the FDA announced its approval of ketamine nasal spray as a treatment for treatment-resistant depression. An addiction-inducing party drug as an FDA-approved antidepressant?! I was skeptical too. At first.
A dire problem
Treatment-resistant depression—depression for which antidepressant medication is not effective—is a dire health concern. In 2016 in the U.S. alone, the National Institutes of Health (NIH) estimates that about 16.2 million adults and 3.1 million adolescents suffered at least one episode of major depression. And that is during only one year. It is estimated that 20% of Americans (about 65 million) will experience a major depressive episode in their lifetimes [1]. Statistics for major depressive disorder do not even take into account bipolar disorder, from which millions more Americans suffer depressive episodes.
The estimates for treatment-resistant depression range, but it is estimated that up to HALF of depression cases are treatment-resistant, leaving many millions of Americans with a debilitating condition that current medication cannot reverse. Furthermore, most antidepressants don’t begin to take effect until several weeks after starting treatment, a period during which there can be increased risk of suicide, particularly among adolescents [2-3]. Our society is indisputably in desperate need of an alternative to existing medication. Ketamine might be a step in that direction.
What is ketamine?
Ketamine was discovered in 1962 and became a widely-used method of surgical anesthesia, particularly during the Vietnam War. It does not suppress heart rate or breathing as much as many other anesthetics do, making it a relatively safe choice for surgery patients. Because of its brief hallucinogenic effects, however, it is no longer the go-to anesthesia method for most surgeries. Today ketamine is most commonly used in veterinary medicine, in pediatric anesthesia, or for patients with airway blockage for whom an inhaled anesthetic is not an option. It is also used as a sedative in emergency medicine. In fact, the World Health Organization gives ketamine a spot on its List of Essential Medicines, a list of the safest, most efficient medications available which, according to the organization, “should be available in health systems everywhere, at all times.”
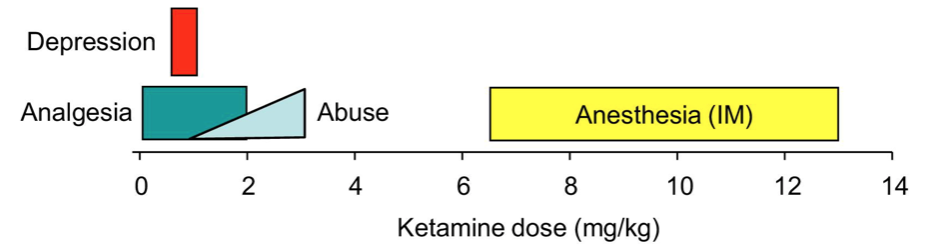
You might not generally associate the rave scene with a patient knocked out in a surgical suite or a List of Essential Medicines. The effects of ketamine can seem paradoxical, but ketamine plays different roles at different doses. (It’s not unique in this regard… a tequila shot might help you brave the dancefloor, but a few too many and you’ll need to be carried home!) At low doses, ketamine acts as a pain reliever with dissociative effects that partiers enjoy. When used for surgery, it is administered at a much, much higher dose to achieve deep sedation. Its use for treatment of depression is on the much lower end of the dosage spectrum.
Ketamine for depression: decades of evidence
Despite its recent FDA-approval, the idea to treat depression with ketamine is by no means new. Almost two decades ago, researchers had begun to string together some lines of evidence that a particular neurotransmitter, glutamate, might be involved in depression. They decided to test whether a drug that acts on the glutamate system might be able to reduce symptoms of depression. The drug they chose to test? Ketamine.
In the first small study done in 2000, ketamine showed promise in reducing depression symptoms [4]. In 2006, a slightly larger study with 18 patients found that one IV infusion of ketamine reduced depression, and did so incredibly quickly; just 110 minutes after the infusion, subjects felt significantly better. While the effect wore off slowly over time, a week after the single infusion subjects were still significantly better than beforehand [5]. And the evidence has continued to accumulate over time. A meta-analysis published in 2012 found that among 163 people receiving a ketamine infusion across several studies, within 24 hours, 25-85% of patients had at least a 50% reduction in one or more measures of depression [6].
Because the recommendation for treatment frequency can be up to twice per week, IV infusions such as those in the study are not particularly ideal for reaching the incredibly large number of people with treatment-resistant depression. The expense of treatment, the time burden on the patient (each infusion takes about 45 minutes), and the repeated needle sticks make even a weekly regimen complicated and costly.
Recently, the pharmaceutical company Janssen patented a ketamine nasal spray called Spravato and sent it through the FDA approval process. Unfortunately, it is still not cheap—a twice-a-week regimen would cost $7000 monthly—but it will be at least partly covered by insurance. The expense does not come from the price of the drug itself, but rather from heavy burdens on doctors’ time and clinic space.
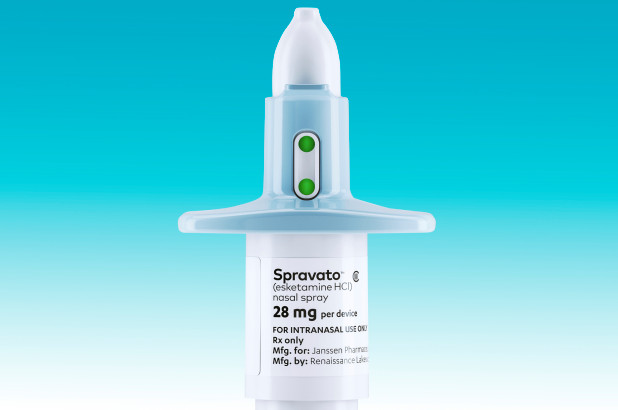
As you might imagine, the nasal spray is not something you can pick up at your local pharmacy. Especially given the stark reality of the current opioid crisis, the FDA would never have approved a nasal spray version of a known party drug for widespread distribution. Spravato will be self-administered, but it will be done so in the presence of a doctor, at a doctor’s office or clinic. The patient will then required to stay in the office or clinic for 2 hours for monitoring to ensure that any dissociative, sedative, or hallucinogenic effects have worn off. The spray never leaves the office or clinic.
How does it work?
Traditional antidepressants, SSRIs, act on the the signaling pathways of a particular neurotransmitter (brain chemicals involved in the communication between two neurons) called serotonin. Ketamine influences a different neurotransmitter, glutamate, the primary excitatory neurotransmitter in the brain.
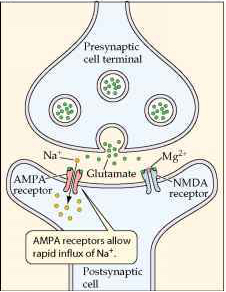
During neural activity, glutamate is released from one cell and binds to receptors on the cell receiving the signal. It can have different effects on the recipient cell depending on which type of receptor it activates. The two glutamate receptors important for ketamine’s action are called NMDA and AMPA receptors. Ketamine binds to NMDA receptors, preventing glutamate from binding as efficiently and therefore decreasing the neuron’s activity. This makes ketamine an NMDA “antagonist”. If ketamine is going around decreasing neurons’ activity, it is intuitive that it could act as a sedative. But how does it work for depression?
It’s not crystal clear. One theory is that at low doses such as those being used for depression, the drug preferentially inhibits the activity of inhibitory neurons, which usually apply a brake on the activity of other neurons. Ketamine decreases the inhibition, releasing the brake, and increasing overall glutamate signaling as a result. This is thought to help relieve depression.
Another theory (not mutually exclusive!) stems from evidence that ketamine convinces neurons to start making more AMPA receptors [7-8] and also increases the density of synapses—the points of communication between two neurons [9]. This would also lead to overall increases in excitatory signaling in the brain. While the details are still being worked out (as is almost always the case in science!), it does seem as if a low dose of ketamine works as an antidepressant by convincing the brain to be more excitable and create more synapses.
How does it feel?
What does ketamine treatment feel like? My brother Matt has been receiving IV infusions of ketamine for several months to treat depressive symptoms from bipolar disorder. They’ve helped him tremendously. I asked him to describe the perceptual experience of an infusion:
It takes about 5-7 minutes after the IV is inserted for me to notice any effects from the ketamine. The first thing I invariably notice is a numbness to my lips, in addition to a general warming of the body, and also sensations of slow, up-and-down movement in my legs and feet. As time progresses, the experience (at least while listening to music) is much like dreaming — the music sort of dictates where you go, what you “see” and the duration of dream sequences (a sequence will end when a song ends, and a new one will start with the next song). I find there is a common theme of movement — nothing nauseating or disorienting, but more like you are on a flying carpet traveling from one dream locale to the next. Although some people report bad experiences, everything has been more than pleasant for me. I’ve experimented with different types of music and every artist seems to bring a unique experience to the infusion — I generally listen to either club trance or psychedelic rock. Anything that you might take recreational drugs to is a good bet.
If I open my eyes during the infusion (and I can, at will, because I am always aware that I am awake) then I find my vision is extremely blurry and my motor skills impaired (for example, switching music can be somewhat difficult to actually do).
After the 45 minute infusion, the 30 minute saline flush begins, and I immediately begin feeling less high. I often fall asleep at this point, because the infusion has made me tired, and there are no longer any dreams/visions holding my attention. If I don’t fall asleep, it’s sort of like being in a meditative state.
Ketamine: A New Hope
Of course, ketamine is not a cure-all. It will not work for everyone with treatment-resistant depression, and even for those it is able to help, it won’t be a perfect cure. Furthermore, its longer term effects are not well-studied. Ketamine does have addictive properties, so while nasal spray access will be tightly regulated, it could become dangerous if all precautions are not heeded.
However, ketamine does seem to hold immense potential for the millions suffering from treatment-resistant depression. It is particularly promising for those with current suicide ideation. Unlike SSRIs which can take weeks to begin to work, ketamine works almost immediately, reducing symptoms of depression within a couple hours after treatment. A study specifically looking at the effect of ketamine on those with current suicide ideation found that the drug quickly and strongly reduced suicidal thoughts [9]. In my opinion, it is this that makes the FDA-approval of ketamine for depression a true game-changer.
References
- Kessler RC, Bromet EJ. The epidemiology of depression across cultures. Annu Rev Public Health. 2013;34:119-38.
- Fergusson D, Doucette S, Glass KC, Shapiro S, Healy D, Hebert P, Hutton B. Association between suicide attempts and selective serotonin reuptake inhibitors: systematic review of randomised controlled trials. BMJ. 2005 Feb 19;330(7488):396. Review. Erratum in: BMJ. 2005 Mar 19;330(7492):653.
- Barbui C, Esposito E, Cipriani A. Selective serotonin reuptake inhibitors and risk of suicide: a systematic review of observational studies. CMAJ. 2009 Feb 3;180(3):291-7.
- Berman RM, Cappiello A, Anand A, Oren DA, Heninger GR, Charney DS, Krystal JH. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry. 2000 Feb 15;47(4):351-4.
- Zarate CA Jr, Singh JB, Carlson PJ, Brutsche NE, Ameli R, Luckenbaugh DA, Charney DS, Manji HK. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006 Aug;63(8):856-64.
- Murrough JW, Iosifescu DV, Chang LC, Al Jurdi RK, Green CE, Perez AM, Iqbal S, Pillemer S, Foulkes A, Shah A, Charney DS, Mathew SJ. Antidepressant efficacy of ketamine in treatment-resistant major depression: a two-site randomized controlled trial. Am J Psychiatry. 2013 Oct;170(10):1134-42.
- Tizabi Y., Bhatti B.H., Manaye K.F., Da Sa J.R., Akinfiresoye L. Antidepressant-like effects of low ketamine dose is associated with increased hippocampal AMPA/NMDA receptor density ratio in female wistar-kyoto rats. Neuroscience. 2012;213:72–80.
- Zhou W, Wang N, Yang C, Li XM, Zhou ZQ, Yang JJ. Ketamine-induced antidepressant effects are associated with AMPA receptors-mediated upregulation of mTOR and BDNF in rat hippocampus and prefrontal cortex. Eur Psychiatry. 2014 Sep;29(7):419-23.
- Li N, Lee B, Liu RJ, Banasr M, Dwyer JM, Iwata M, Li XY, Aghajanian G, Duman RS. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science. 2010 Aug 20;329(5994):959-64.
- Ionescu DF, Swee MB, Pavone KJ, Taylor N, Akeju O, Baer L, Nyer M, Cassano P, Mischoulon D, Alpert JE, Brown EN, Nock MK, Fava M, Cusin C. Rapid and Sustained Reductions in Current Suicidal Ideation Following Repeated Doses of Intravenous Ketamine: Secondary Analysis of an Open-Label Study. J Clin Psychiatry. 2016 Jun;77(6):e719-25.
- Glue P, Gulati A, Le Nedelec M, Duffull S. Dose- and exposure-response to ketamine in depression. Biol Psychiatry. 2011 Aug 15;70(4):e9-10; author reply e11-2.

You must be logged in to post a comment.