 June
18
June
18
Tags
Left over right: Communicative auditory processing from mice to men

The terms “left” and “right”, if not used to give directions, are often evoked to describe a preference. You might prefer to kick with your right leg, or throw with your right arm, or lead with your right foot on your skateboard or surfboard. Political convictions often lean left or right. And then there’s handedness, arguably the most widely used and universal form of left-right preference.
The brain, too, exhibits certain left-right biases. While things like artistic abilities and math skills aren’t as strictly lateralized to a single brain hemisphere as some articles on the internet might lead you to believe (see a previous NeuWrite post about the origins of the “left-brained”/”right-brained” myth), there are undeniable asymmetries in how the human brain performs certain functions.
One such function whose lateralization is particularly well-known and well-studied is language. This dates back to 1865 when the French neurologist Pierre Paul Broca presented his findings of patients with brain lesions confined to a particular region of the frontal cortex in the left hemisphere – now appropriately named “Broca’s area” – who had lost the ability to speak [1]. Ever since, we’ve known that the left hemisphere holds privileged access to our verbal abilities, although fascinating exceptions have also emerged (for instance, rare instances of right-lateralization that are only found in left-handed individuals [2]).

While scientists don’t fully understand why this functional asymmetry for language exists, it is often discussed with a tinge of anthropocentrism. There has been substantial research into whether other animals show any early signs of left-right biases in the brain, and the general sense is that while some cases exist, they are less evident than in humans. For instance, although it has been a topic of much debate, it seems as though non-human primates lack the consistent left-lateralization of Broca’s area that is characteristic of humans [3]. This has led many to believe that this is a uniquely human feature that may have caused or been a consequence of the birth of human language in our evolution. This stands in contrast to the primate homolog of Wernicke’s area – another brain region important for language in humans, but particularly for language comprehension, as opposed to production (like Broca’s). Certain features of Wernicke’s area have been found to be left-lateralized in great apes as well [4,5], suggesting that a left-biased system for listening to communicative sounds may have evolved before that for producing those sounds.
As a matter of fact, there are a surprising number of examples of hemispheric asymmetries related to communication, if not language per se, in non-human animals. Even mice exhibit certain left-right biases in terms of auditory processing, particularly in the context of maternal behavior. And guess which side of the brain seems specialized for processing sounds produced by other members of their species?
Left lateralization in mouse auditory cortex
While left-lateralized processing of communication sounds had already been observed in non-human primates (e.g. [6]), this might not have been surprising given their relatively close evolutionary relationship to humans and that, while short of language, they utilize a sophisticated and expansive repertoire of vocalizations for communication. It was arguably much more surprising, however, when Gunter Ehret in 1987 identified leftward biases in vocalization processing in ordinary house mice [7]. While mice frequently produce ultrasonic vocalizations that are beyond the range of human perception, a particularly salient category of vocalizations that elicit a stereotyped behavioral response are those of crying mouse pups. For instance, if a young mouse pup is displaced from its nest, it will elicit an ultrasonic cry for help that experienced mouse mothers will recognize and respond to by retrieving the pup and bringing it back to the nest.
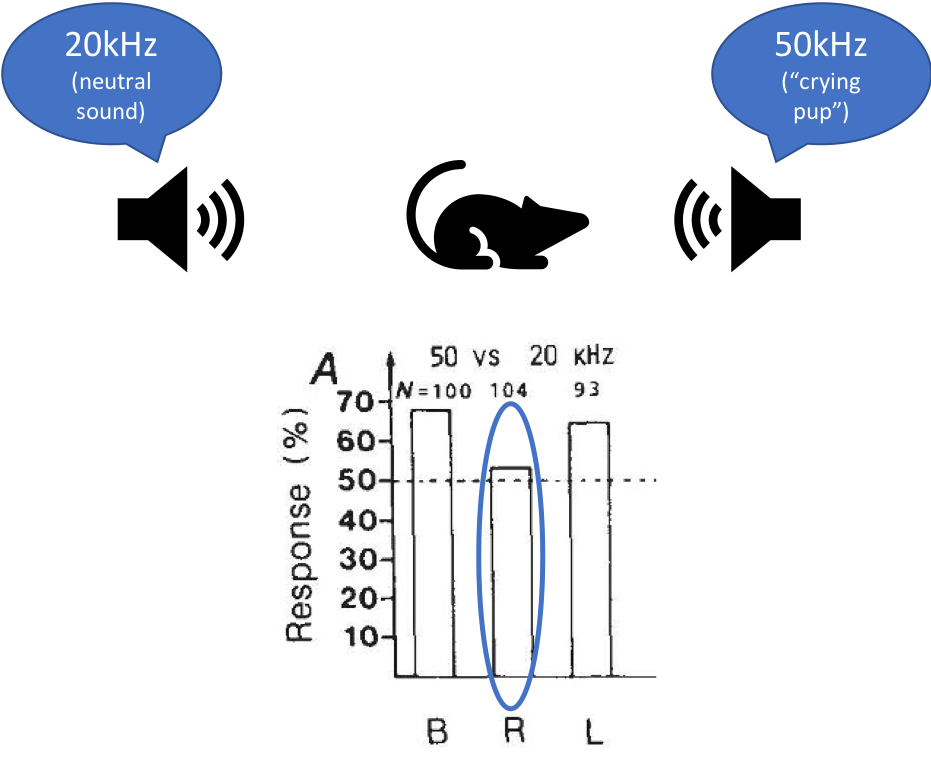
In Ehret’s experiment, mouse mothers were placed onto a running board with speakers on either end playing one of two different sounds, one of which resembled pup cries. Ehret then tested their preference for approaching the speaker that played the artificial pup cries under three conditions: when their left ear was plugged, when their right ear was plugged, or when both ears were open. Fascinatingly, while the mouse moms preferentially moved towards the speaker playing artificial pup calls when both ears or just the right ear was open, this preference disappeared when their right ear was closed. Since the left hemisphere of the brain primarily processes sounds coming in through the right ear and vice versa, this would suggest that auditory processing in specifically the left hemisphere of the brain is needed for the mouse moms to recognize the cries of their discomfited pups. Interestingly, this right ear/left brain bias for pup cries was absent in inexperienced female mice who had not learned the association between these high-frequency sounds and distressed pups. If they were trained to approach the speaker playing artificial pup calls for a reward, either ear was sufficient for recognizing the sounds and moving in the direction of the pup vocalization sounds. Thus, it appeared that left-lateralized processing of those sounds is not inborn but instead depends on the learned communicative salience of those sounds.
How is it that the exact same auditory stimulus can be processed preferentially by the left brain in mouse moms but not in inexperienced females? More recent studies have offered evidence that oxytocin – a brain hormone closely associated with social behaviors such as pair bonding and maternal care – plays a key role and displays its own left brain bias. Recall from Ehret’s research that the inexperienced females, in contrast to the experienced moms, had to be specifically trained to respond to the pup cries. But Marlin et al., (2015) found that if the inexperienced females were given a boost of oxytocin and a few days to observe experienced moms in action, they started bringing distressed pups back to the nest at a higher rate (you might say that they became “better moms”). Moreover, the left, but not the right, auditory cortex was necessary for the moms to retrieve crying pups [8].
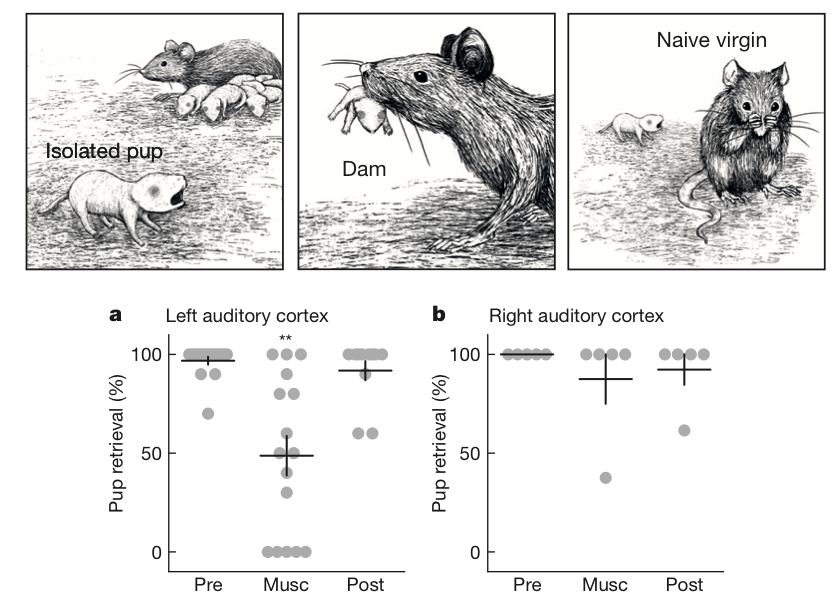
Since oxytocin and the left auditory cortex seem to both be important for the maternal behaviors evoked by pup vocalizations, is there something different about oxytocin processing in the left hemisphere? The researchers found that the answer was a resounding yes; female mice (regardless of maternal experience), but not male mice, had more neurons with the oxytocin receptor in the left auditory cortex than in the right [8,9]. Moreover, oxytocin signaling caused long-lasting changes in how neurons in the left auditory cortex responded to the sound of pup vocalizations. When inexperienced females got a boost of oxytocin, their responses in the left auditory cortex to the sounds of pup cries more closely resembled those of the experienced moms [8]. Thus, oxytocin in the left auditory cortex in particular is important for processing and responding to pup vocalizations in female mice.
But this left brain bias for processing communicative sounds extends beyond maternal behaviors in mice. In fact, male mice also show left-biased auditory processing of vocalizations. A recent study found that more neurons in the left than right auditory cortex in male mice were activated by recordings of mouse vocalizations, whereas more neurons in the right auditory cortex were activated by frequency sweeps (rapid increases or decreases in pitch) [10]. This is starkly reminiscent of findings in humans; although language processing is left-lateralized, things like pitch and intonation seem to be biased towards the right (hence why the right hemisphere has been reported to be especially responsive to music [11] ). The researchers also found that neurons in the left hemisphere were activated preferentially by downward sweeps from high to low frequencies – a particularly common feature in mouse vocalizations – whereas neurons in the right hemisphere were more variable in their sweep direction preferences. However, normal auditory experience was required for vocalization processing to become left-lateralized [10]. This is similar to Ehret’s findings that leftward bias was absent in inexperienced female mice and the fact that leftward bias of oxytocin receptors in the left auditory cortex is absent early in development [9].

These findings, combined with the extensive work on lateralization in humans and non-human primates, suggest that there has been something special about the left hemisphere for processing the sounds of one’s own species for tens, if not hundreds, of millions of years in our evolutionary history. But this begs a number of important and fascinating questions. First, why does this lateralization occur? The fact that it appears to have emerged so long ago in our evolutionary history and stuck around suggests that it must be serving some useful function. The most widely accepted hypothesis so far holds that a “division of labor” approach by the brain may improve efficiency. Since auditory processing, particularly in the context of communication, requires processing rapidly changing sounds (high temporal resolution) at a wide range of frequencies (high spectral resolution), dividing these processing priorities between brain hemispheres is thought to be advantageous for maximizing both high temporal and spectral resolution [12,13].
But then, how does this lateralization come about, and why in the left brain in particular? While these various animal studies show that lateralization isn’t present at birth but rather emerges later in development, the fact that it emerges so consistently across animals (and even species), and always in the left hemisphere, suggests that the left brain is in some way genetically predisposed to serve this special role. In theory, if improved efficiency were the only goal, there could be some mice whose vocalization processing is concentrated in the left auditory cortex and others in the right, and equal proportions of right- versus left-lateralized language processing in humans. But instead, ~90% of humans show left language bias, and right lateralization hasn’t been described in mice. Likely this has to do with the particular genetic programs that are involved, which might themselves operate in a hemisphere-specific manner rather than flipping a coin. This is biologically plausible; there are a number of genes which are known to be expressed to different degrees on different sides of the brain and/or body [14], although specific genes that govern the left lateralization of vocalization processing have yet to be identified. In the meantime, we can marvel over the fact that some biological force, many millions of years ago, determined that something would be special about the left side of the brain for listening and responding to the sounds of one’s own species.
References:
1 Güntürkün, O. and Ocklenburg, S. (2017) Ontogenesis of Lateralization. Neuron 94, 249–263
Left/right image: https://images.ctfassets.net/qnesrjodfi80/7ljFo1dCnYoICwQUAaEeiy/e027b10316be9a04aab6411d69065f7d/left_vs_right_16x9.jpg?w=1280&h=720&q=50&fit=fill
Broca’s/Wernicke’s: https://en.wikipedia.org/wiki/Wernicke%27s_area#/media/File:BrocasAreaSmall.png

Pingback: Nyheder2020juni17 - bionyt.dk
Pingback: Left-v-Right: How do two groups of people view the same evidence and facts but have strikingly different opinions? – John N. White