 July
20
July
20
Tags
Genomes, Circuits, and the Roundworm: C. Elegans as a Model Organism
Caenorhabditis Elegans, Who Are You?
What do you consider easier, putting together a 10,000 piece puzzle, or a 100 piece one? The human brain, with its vast interconnections and seemingly innumerable neurons, has proved to be a challenging starting point when studying the cause for neurological disease. As a result, much of pharmaceutical research is done with mice, but even at the level of a mouse, disentangling the cell-to-cell circuitry underlying brain function can lead to similar hurdles as with the human brain: there are still too many neurons to easily manage!
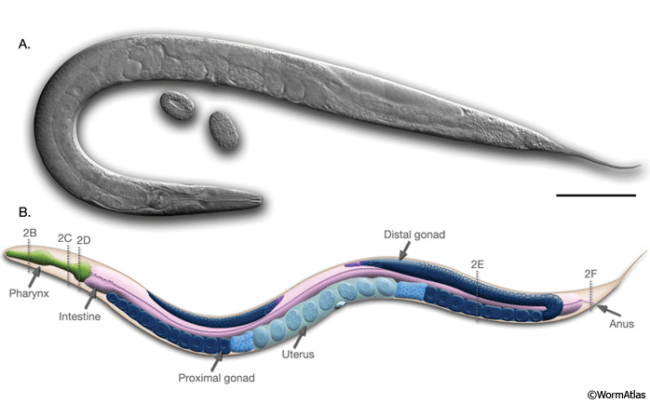
Anatomy of an adult C. elegans hermaphrodite. A. DIC image of an adult hermaphrodite. Scale bar 0.1 mm. B. Schematic of anatomical structures.
Over 50 years ago, Dr. Sydney Brenner and colleagues introduced Caenorhabditis elegans (C. elegans) as a model organism, with the idea that extremely simple organisms can provide a less cluttered workbench from which to study biological processes in the living nervous system. The soil nematode has since wormed its way into laboratories around the globe, where it is used as a model to study stress, development, reproduction, and the central nervous system, to name a few.
How can a tiny organism, about 1 mm in length, end up telling us so much about the biology within us? Let’s look at some of the benefits of using C. elegans, as well as some limitations, that will illustrate why it rose and remains a valuable research tool.
Cheap and Easy Maintenance
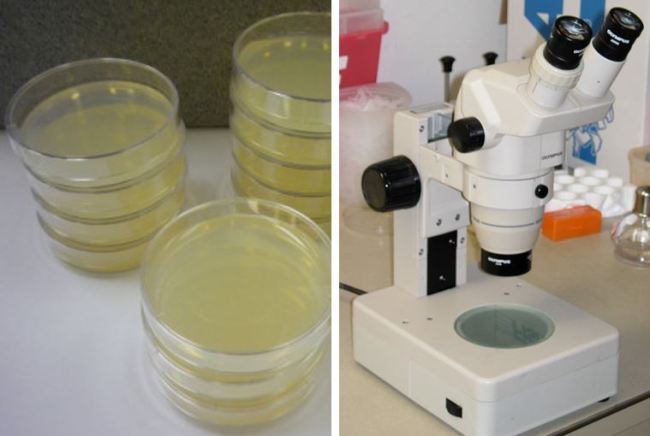
C. elegans Maintenance. (Left) C. elegans petri dishes. (Right) Microscope typically used for viewing.
The size of C. elegans makes their housing cheap and simple. They can be cultivated in large quantities, up to ~10,000 of them in a single petri dish. These petri dishes are not difficult to prepare; all that’s needed for a happy C. elegans population is a layer of gelatinous Nematode Growth Medium (NGM) and a bacterial food source. This food source is typically E. coli bacteria that have been extracted from a liquid culture. Once the bacteria have settled overnight, a fancy tool called the “worm pick” can be used to move worms from a source plate onto your prepared dish. Your C. elegans population will now reproduce and eat until its food source has expired. But don’t worry! Before that happens, you can move a couple of your worms onto a new dish, and the cycle can begin anew. Its inexpensiveness certainly helps when funding a laboratory, and it takes only a few hours to train a new student to maintain a population of C. elegans.
Rapid Life-cycle
C. elegans has two sexes: hermaphrodites and males, with hermaphrodite C. elegans comprising the majority. Their self-fertilizing production process makes them a valuable tool for genetic study. A single C. elegans hermaphrodite produces more than 300 offspring in their lifetime, each containing a similar genetic code as their “parent”.

C. elegans Lifespan. Unfavorable conditions lead to a dauer state.
Each egg takes about 3 days to hatch, and with enough food and not too much overcrowding, lives up to 2 weeks as a fully matured adult. To get there, however, it must undergo four different larval stages (L1-L4) that last 8-12 hours each. If crowding, starvation, or other unfavorable environmental stressors strain its development into L2, the organism goes into an arrested state called “dauer” (circled in red above). This is in stark contrast to other laboratory organisms that starve without access to food! C. elegans in dauer can continue their development soon after being re-exposed to favorable conditions, saving your experiment if you happen to forget to move them onto a fresh, prepared plate.
The predictable and discrete developmental stages of C. elegans is a boon to scientists studying the maturation of the nervous system. C. elegans enables scientists to answer questions related to the timeline of cellular connectivity, as well as what occurs to these connections when different stressor, gene, and molecular manipulations are introduced to the organism at each of these stages. However, for these studies to be possible, you’d need to see what is happening inside the living organism as it ages.
Watch C. elegans in motion here!
Transparency
C. elegans are naturally transparent. Observations of internal structures, such as its digestive and reproductive systems, can be done in real time under a microscope. Studying a gene’s influence on the arrangement of cells in the organism can be done by tracking cells with different genetic codes and following their lineages, a task not easily accomplished in real-time with other animal models (I’m still waiting for live, transparent mice).
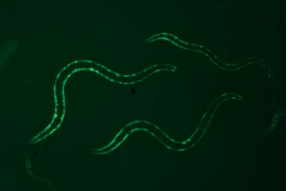
Mutant C. elegans. This mutant has been genetically altered to express Green Fluorescent Protein (GFP) in specific cell muscle.
By now, several stress and developmental molecular pathways are defined in C. elegans. Multiple labs have contributed to the development of C. elegans strains that express Green fluorescent protein (GFP). When combined with fluorescence measurement techniques and the organism’s transparency, C. elegans can serve as model to tests for environmental causes of disease.
Sequenced Genome
C. elegans was the first multicellular organism to have its genome completely sequenced and revealed. The organism’s DNA is composed of six pairs of chromosomes, altogether encoding more than 19,000 proteins– only slightly fewer than the number in humans! This means that any motivated scientists can assay any of the genes present in C. elegans. Finding the function of more than half of its genes is an ongoing yet feasible process due to the reasons described above. In addition, a substantial number of genes (35%) have been found to have equivalent genes in humans, some of which have been linked to diseases such as Parkinson’s. A sequenced genome means that regions in their DNA are now prime for manipulation, and comparisons between mutant and wild-type (the typical form of the species) variants could reveal enhancements or suppression of phenotypes (organism characteristics) associated with that gene.
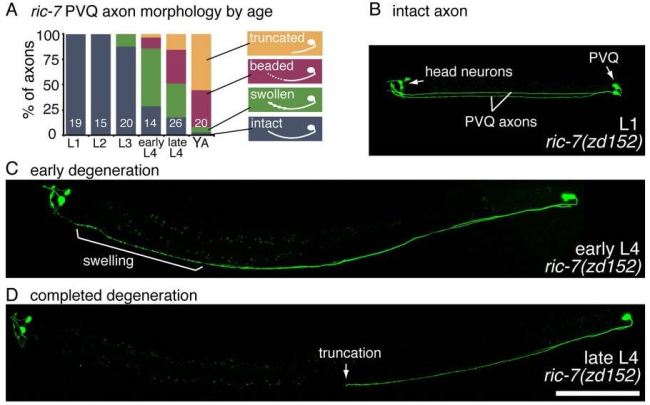
Mutant used to study neuron degeneration. When the ric-7 gene is altered in a certain neuron type (PVQ), mitochondria become absent in these cells, causing axons (connections) to quickly degenerate over time.
Fully Mapped Circuitry
Finally, another unique characteristic of working with C. elegans is their consistent cell count. A typical worm contains 959 cells at adulthood, and movement throughout development is constant. Understanding behavior in the context of a network comprised of millions of brain cells that are constantly changing connections is a gargantuan task. However, with enough probing of C. elegans cell-to-cell circuitry, one can establish a predictable link between behavior and neural anatomy. Only about a third of the cells in C. elegans can be considered equivalent to neurons in function, providing a tractable and consistent set piece from which to study how the brain manages every aspect of an organism.
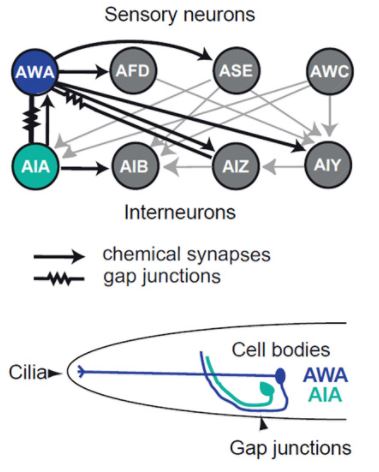
A neural circuit for chemotaxis (chemically-guided movement). (Top) Diagram showing the different connections between olfactory neurons. (Bottom) Schematic of their location within C. elegans.
What Else Could be out There?
Sometimes, to study a complex system, it may be better to start with something simpler. C. elegans is easy to manage, and every single one of its 959 somatic cells is visible under a microscope due to its transparent body. A fast life-cycle and a known genetic code makes C. elegans the ideal balance between complexity and manipulable. Their behavior is easily measurable, and its ability to sense noxious chemicals in its surroundings for avoidance highlights the organisms utility in environmental research. Keep in mind, the next time you go to the clinic, an organism with no eyes and limbs could have served as the basis for studying the effects of your prescribed medicine. In nature, even the smallest organisms can be powerhouses for research. Who knows, with multiple new species discovered every year, other organisms from big to small could be out there in the wild, currently unnoticed, but nonetheless with the hidden potential to unlock the mysteries of what makes us human.
Sources:
Figure 1: WormAtlas
Figure 2: WormClassroom
Figure 3: Shawn Xu Lab
Figure 4: GFP C. elegans Protocol
Figure 5: Rawson et al. 2014
Figure 6: Larsch et al. 2015
Figure 7: Wikipedia

Pingback: Dopamine is NOT your brain’s reward chemical | NeuWrite San Diego