 September
05
September
05
Tags
“I see it, I like it, I want it, I got it”
I would never have thought that an Ariana Grande song could lend itself to talking about an interesting avenue of neuroscience research. Yet the catchy phrase repeatedly featured in her most recent hit song “7 rings,” “I see it, I like it, I want it, I got it,” implicitly highlights the relationship between liking and wanting, two psychological concepts which are surprisingly quite separable at the level of the brain.
Though the linear flow of the song lyrics may suggest that you like something, and then want it, in reality one does not have to precede the other. Think about it – just because you like something, that doesn’t necessarily mean you’ll want it. On the flip side, just because you want something, that doesn’t necessarily mean you like it. Rather, liking and wanting are processes that can co-exist and drive distinct behaviors.
Before I go on, let’s pause here and get into the neuropsychological definitions of these words, for differences in the brain circuitry underlying liking, reward processing, wanting etc. can be hard to study without operationalizing the terms. Though there is a great deal of overlap in the colloquial meanings that the words “liking” and “wanting” evoke, in the context of this piece, “liking” specifically refers to brain processes that underlie perceiving something as pleasurable, whether consciously or not. “Wanting” refers more to brain processes that underlie motivation to obtain a rewarding thing, and in conscious form, is directly linked to having a cognitive, declarative goal (1).
To give a personal example of the interplay between liking and wanting, among my many guilty pleasures is eating Cheetos. I never buy Cheetos for myself when I go grocery shopping and I never explicitly seek them out in any other context because I am set on maintaining healthy eating habits. However, any time Cheetos are in a bowl in the same environment as me, whether that be at a party or conference luncheon, I will more often than not pop a few in my mouth and actually derive pleasure from the experience of eating the Cheetos – the crunch and taste and everything about them. So while I can honestly say that I don’t want Cheetos in my life, it’s clear to me that I like them. I’ll even perform the classical orofacial behaviors that animals display when they are tasting something they like, such as when I lick my orange-dusted fingers and lips (Fig. 1).

Figure 1: Liking and disgust-related orofacial behaviors are innate and conserved across species (adapted from reference 2).
Objective liking of food, water, sex etc. is easier to study in a laboratory setting even though in the case of our cognitively advanced species, “liking” is often a conscious and variable experience of pleasure (subjective liking). In fact, one of the characteristics that sets us apart from other species is the fact that our repertoire of things we “like” extends well beyond what we share with other species into aesthetic, moral, and cognitive domains (e.g. literature, music, ethics, money, etc.). Thus, relying on human studies to understand brain processes underlying liking can be tricky: Techniques available for studying humans are limited to making correlational conclusions about how behavior relates to brain activity patterns. Furthermore, there is an inherent problem of a person giving a subjective rating for how much they like something. Thankfully, certain liking behaviors are largely innate, as well as conserved across species (Fig. 1). For instance, taste reactivity patterns to sweets (e.g. rhythmic and lateral tongue protrusions) are homologous in human infants, primates, and rodents in the same way that gaping, forelimb flails, and head-shaking in response to bitter tastes are innate and conserved behaviors (2). Thus, these types of behaviors can be used as objective metrics to quantify how much an animal likes something, and whether manipulation of candidate brain regions affects how much an animal demonstrably likes something.
These behavioral observations suggest an important role for brain processes underlying liking in evolutionary fitness and survival. Indeed, Kent Berridge and colleagues who have devoted a significant portion of their careers to uncovering the dissociable neurobiology of liking vs. wanting have appropriately termed pleasure as “evolution’s boldest trick.” Appropriate, because if organisms didn’t like eating, drinking, having sex etc., most species would almost certainly not make it very long in the world. With all that in mind, perhaps it is not entirely shocking that there are “hedonic hotspots” in the brain.* Hedonic hotspots are brain regions that are termed so because they activate in response to sensory pleasures such as food intake and sex, and in the case of humans, also the aforementioned higher types of pleasures. A fascinating implication of having hedonic hotspots in our brains is that the hedonic impact (i.e. the niceness vs. nastiness) of something out in the world is not an inherent property of that thing, but rather a function of the processing that is going on in these hedonic hotspots and the brain circuitry they are embedded in. And if this is so, by manipulating activity in these hedonic hotspots, can we alter how much something is liked?
* Hedonic comes from the ancient Greek word hedone, meaning pleasure.
A prominent brain region that houses a hedonic hotspot is called the nucleus accumbens (NAc) (Fig. 2). Studies have demonstrated that by activating opioid receptors in the hedonic hotspot subregion of the NAc, one can amplify the positive hedonic impact of something, or more simply put, make a mouse “like” something more. One such study did as follows: Fifteen minutes after microinjecting a synthetic compound that activates opioid receptors into the NAc hotspot, investigators recorded the affective facial and body reactions of rats as they received sugar solution via an oral cannula. They observed that the positive hedonic reactions of rats more than doubled as compared to when rats were microinjected with a control vehicle that doesn’t affect NAc activity (4).
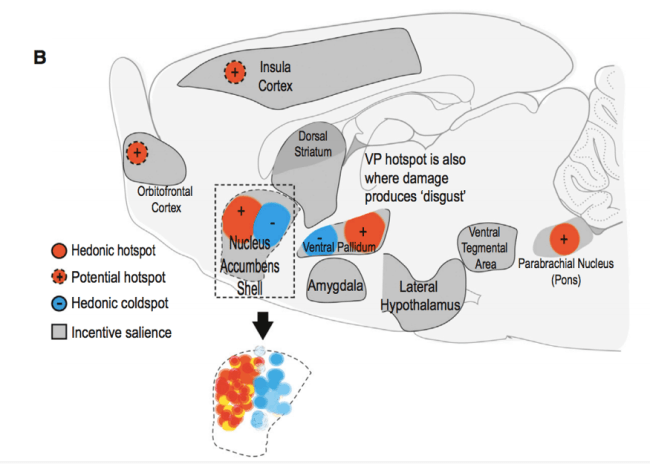
Figure 2: Side view of a rodent brain schematic. Two brain regions that have stood out as hedonic loci in the brain are the nucleus accumbens (NAc) and the ventral pallidum (VP) depicted as housing both hedonic hot and coldspots (adapted from reference 3).
Multiple different signaling pathways run through every brain region; thus, while opioids influence processing in the NAc, so can other signaling molecules. One such neurotransmitter is called dopamine. Dopamine signaling is thought to heavily influence motivational processes in the brain, and dopamine’s role in the NAc in particular has been found to be important for the expression of “wanting” behaviors. In contrast to the previous study, enhancing dopamine signaling in the NAc of mice did not increase positive hedonic reactions elicited by sugary solution consumption (i.e. sugar “liking”). However, enhancing dopamine signaling in the NAc did, in the absence of actually receiving a sugar reward, motivate the mice to perform actions to earn a sugar reward (5).
Another study with similar findings used a more general approach: These investigators worked with mutated mice that did not express normal levels of dopamine transporter (DAT). The role of DATs is to clear away dopamine from the areas where neurons interface with one another for communication. So these DAT knockout mice had 70% elevated levels of synaptic dopamine which meant that DA signaling was increased in these mice. The investigators observed that the DAT knockout mice left the start box more quickly than control mice with no mutation, required fewer trials to learn the task, resisted distractions better, and proceeded more directly to retrieve the sweet reward (6). In a nutshell, affecting dopamine signaling seems to influence the “wanting” processes that underlie reward-related behaviors without influencing the hedonic impact of the reward.
These dopamine studies are quite intriguing in light of a subset of Parkinson’s patients who struggle with pathological gambling, hypersexuality, and/or compulsive eating due to a treatment that affects dopamine brain circuits. One of the most notable features of Parkinson’s is the death of dopamine neurons in a part of the brain called the substantia nigra. To compensate for the death of this neural population, a common treatment for Parkinson’s is to give patients the precursor to dopamine, L-DOPA. Given all of the studies that implicate dopamine in motivational processes, it is not entirely unexpected that a nonendogenous increase in dopamine can in some cases lead to “wanting” to perform the behaviors listed above. But though these patients are performing more behaviors that seek out gambling, sex. etc. during L-DOPA treatment, it is still an open question whether they actually like gambling more than they used to.
Interestingly, when Parkinson’s patients who received L-DOPA treatment and developed hypersexuality observed a mix of visual stimuli including images of food, money, gambling, sexually suggestive behaviours, and neutral cues like nature scenes, the activity patterns of their hedonic hotspot regions did not correlate with their report of how much they liked the various stimuli (7). While this fMRI study is not perfect in design and execution, it is still interesting to think about how the dissociable brain circuits of “liking” and “wanting” may leave us vulnerable to pathologies where we perform excessive behaviors to obtain something even when we don’t necessarily like some facets of the object of pursuit more.
Taking a step back, what could be the utility of evolution resulting in brain circuitry that dissociates “liking” and “wanting”? Kent Berridge and colleagues posit that in a world full of a whole host of things we like, it is useful for our brains to be able to help us prioritize what is most advantageous in the present by utilizing wanting and motivational processes. If a person is famished, they’re most likely not going to be interested in other activities they like until they are full, and will instead beeline to their favorite eatery. And with that, I am headed to lunch.
Ege Yalcinbas is a 4th year student in the UCSD Neurosciences Graduate Program. In her spare time away from lab, she likes puzzling, goofing off with her friends, performing at concert venues and/or on stage, and loving her cat.
Referenced Publications:
- Affective neuroscience of pleasure: reward in humans and animals. Psychopharmacology (Berl). 2008 Aug;199(3):457-80. 10.1007/s00213-008-1099-6.
- Measuring hedonic impact in animals and infants: microstructure of affective taste reactivity patterns. Neurosci Biobehav Rev. 2000 Mar;24(2):173-98.
- Pleasure systems in the brain. Neuron. 2015 May 6; 86(3): 646–664. 10.1016/j.neuron.2015.02.018.
- Hedonic Hot Spot in Nucleus Accumbens Shell: Where Do-Opioids Cause Increased Hedonic Impact of Sweetness? J Neurosci. 2005 Dec 14;25(50):11777-86.
- Intra-accumbens amphetamine increases the conditioned incentive salience of sucrose reward: enhancement of reward “wanting” without enhanced “liking” or response reinforcement. J Neurosci. 2000 Nov 1;20(21):8122-30.
- Hyperdopaminergic Mutant Mice Have Higher “Wanting” But Not “Liking” for Sweet Rewards. J Neurosci. 2003 Oct 15;23(28):9395-402.
- Sex, drugs and Parkinson’s disease. Brain. 2013 Feb; 136(2): 371–373. 10.1093/brain/awt022.

You must be logged in to post a comment.