 October
08
October
08
Tags
It’s about time
About a day
To you, Earth probably seems like a fairly stagnant entity. Sure, we are currently dealing with rising global temperatures as well as carbon dioxide (CO2) levels, but in your day to day world, those changes probably seem quite small. Over the history of life on Earth, however, so many things that we normally take for granted have changed drastically, such as the temperature, nutrient levels, atmospheric oxygen levels, and even Earth’s magnetic field. But one thing has stayed consistent during the entire time life has existed on this planet: that is the seemingly trivial fact that every single day, the sun rises and the sun sets. It is pure clockwork; life can always count on the fact that as Earth spins about its axis while orbiting the sun, we will always have a consistent night and day.
So what did very early life forms on this planet do? They “learned” how to predict when there would be sunlight and when there would not. From as far back as we know, living organisms were able to alter their biology in a rhythmic pattern which matched the cycle of the rising and setting sun each day. These patterns are called circadian rhythms, which is derived from the Latin roots circa, “about” and diem, “day”, about a day. This circadian rhythm allows life to make the most out of its situation. Let’s take plants as an example. You are probably aware that they use photosynthesis to produce energy to stay alive. They actually capture the incoming energy from the sun’s rays to produce a usable form of energy for them to live on and grow. But what about night-time when the sun is no longer shining? If plants were always counting on sunlight, they would die not long after the sun sets, as there would be no incoming energy for them. Instead, plants have a circadian rhythm which allows them to anticipate nighttime and store energy to get them through the night until the sun is shining once again the next day.
Plants are not the only living things that have circadian rhythms; most living things do [8]. And in fact, each cell in a multicellular organism such as yourself has its own circadian rhythm. This means you can cut out a cell from a plant or animal, and if you can keep it alive in a dish, that cell will continue to have 24 hour cycles, even in the absence of light. In this article, I will start by discussing how circadian rhythms work in a single cell, then I will discuss how these rhythms are synchronized throughout a multicellular animal such as yourself. Finally, I will discuss some of the correlations that have been discovered between circadian rhythms and health and disease.
How do cells keep track of time?
Every cell in your body has its own circadian rhythm. Take some random cell in your liver, which never sees the light of day… it has a 24 hour rhythm by which its physiological functions fluctuate. And this would continue to be the case even if you removed that cell from the body. So how does this work? It’s actually a fairly complicated process involving many different proteins and genes, but the concept is very simple. The cell uses what is called a negative feedback loop. Before we dive into the specifics of circadian negative feedback loops, I’ll start with a more familiar example. Imagine there is an ecosystem where the only two animals are rabbits and coyotes (who eat rabbits). If the number of coyotes in this system starts to increase, they will reduce the number of rabbits and, therefore, the number of coyotes will drop since there is less food to sustain a large population. But then as the number of coyotes goes down, the rabbit population will be able to grow (since there are fewer predators) and therefore the number of coyotes will go back up. This is a highly simplified example, but you get the idea. The more coyotes there are, the fewer coyotes there will be in the near future. And the fewer coyotes there are, the more coyotes there will be in the near future.
A brief note on how proteins are made

You may be aware that proteins are responsible for performing countless duties in the body. There are proteins, like collagen, that help make up the structure of your body, proteins like insulin that act as hormones, and the list goes on and on; proteins are very important for your body to function. But where do proteins come from? And the answer is not from eating meat and beans. When you eat foods high in protein your body breaks them down into the parts that make them up (called amino acids). Then your body makes its own proteins, which are made of a specific sequence of parts (amino acids) that give the protein the correct shape to do its necessary function, like provide structural support for the body. So how does the body know how to string together the correct parts to make the correct protein? That is where genes come into play, which are just specific chunks of DNA. The DNA, which is kept inside the nucleus of the cell, is essentially like a recipe book. It stores all of the information necessary to make the individual. But just like in a recipe book, in order to get the final product, such as chocolate cookies, you must follow that recipe and put in the right ingredients in the right order. But the DNA is very important, it’s the cells only copy of all the genetic information, which is why it’s safely kept inside the nucleus. So instead of just using the recipe to make the cookies if you will, instead what happens is the recipe is copied (in a process called transcription) and that copy leaves nucleus (into what is called the cytosol) where it can be used to make the final product, aka a protein. The “copy” of the gene which leaves the nucleus to be “read off” to make the protein is called mRNA. The process of using the mRNA recipe to make the actual protein is called translation. So the more a gene is transcribed into RNA and made into a protein, the more of that protein there will be in the cell. If something were to turn off transcription of a gene that would result in less protein being made.

Now back to negative feedback loops. Many processes in the body utilize these negative feedback loops, which allow the body to maintain consistency in all sorts of things like temperature and nutrient levels. Now that we understand negative feedback loops in the case of coyotes and rabbits, let’s briefly talk about how circadian cycles work. If you really want to get into the nitty gritty details of this, I’d recommend this article. At the heart of the circadian clock is what’s called a transcription–translation feedback loop (TTFL), which takes approximately 24 hours to complete. I will focus on the mammalian circadian clock, but it works very similarly in other organisms as well. In the case of the circadian clock, one particularly important protein is called CRY. The gene for CRY is transcribed in the nucleus, and the RNA moves into the cytosol where the protein CRY is made. Then as the levels of the protein CRY increase in the cytosol, CRY travels back into the nucleus where it acts as a “transcription factor”, which is essentially a switch to turn off the transcription of the CRY gene. When CRY turns off transcription of the CRY gene, the amount of CRY protein in the cytosol decreases. This results in less CRY to be able to turn off transcription which allows more CRY gene to be transcribed. The more CRY there is, the less CRY is made and vice versa, just like how increasing levels of coyotes results in fewer coyotes. In the case of CRY, the entire cycle takes about 24 hours. While CRY is just one of the many players responsible for the 24 hour circadian rhythm in a cell, the point here is that this cyclic nature of protein synthesis takes a specific, biologically relevant amount of time. Processes like this are able to serve as a mechanism by which a cell can maintain a 24 hour rhythm.
Body-wide synchronization

It’s one thing for every cell in the body to have 24 hour-cycling functions, but in order for the whole body to work together correctly, the cells all have to be synced up. You wouldn’t want a cell in your kidney to think it’s nighttime while a cell in your lung thought it was daytime. There are actually many ways by which the body forces the cycles of each of the cells to be in sync with each other, such as hormones like insulin that circulate throughout the body. But it is not enough just for all of the cells to be in sync with each other, they also must be in sync with actual night and day time. Have you ever had jet lag? That awful feeling is caused by the misalignment of the night and day cycle of the time zone you travelled to and the night and day cycle your body was used to from home. Your body will eventually shift to the new time zone, but it’s not a fun process. In order for the body to be in sync with night and day, it must know when the sun is out. There is a part of your brain, called the suprachiasmatic nucleus (SCN for short) that is known as the “master regulator” of the circadian rhythm of the body. This very small structure sits right above the optic chiasm, which is where the two nerves coming from your two eyes cross each other. This is a prime location for the SCN because it is able to receive light information coming in from the eyes. In fact, there is a special type of light receptor in the eye, in addition to rods and cones, that are solely responsible for telling the SCN if it’s light out. The SCN is then able to relay this information to the rest of the body allowing each mini clock in all of your cells to be in tune with each other and with the cycle of night and day. That is, if everything is going as it should.
Circadian rhythms: they’re more important than you might think
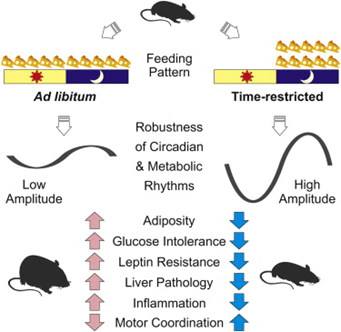
You may be wondering why circadian rhythms matter for humans at all. Nowadays it can be “daytime” whenever we want due to lightbulbs and screens. But, in fact, we are learning more and more about how important circadian rhythms are in health and disease. Circadian disruptions have been linked to many different disorders including cardiovascular disease, diabetes, obesity, cancer, depression, bipolar, schizophrenia, attention deficit [5], Alzheimer’s disease, and Parkinson’s disease [6]. For the remainder of this article, I will focus on the link between the circadian rhythm and metabolic disease, including diabetes and obesity. Early on, scientists looking to study metabolic disease were faced with the need to create an animal model of the disorder. They developed what is called the high-fat diet model, which, as you may guess, involves feeding rodents a diet high in fat and carbohydrates in order to cause them to become obese and develop diabetes. And it works; if you feed a mouse a high fat diet, they will begin to gain weight and develop metabolic disease. But the story isn’t quite so simple. Scientists who were studying this phenomenon noticed that, unlike normally fed mice who sleep through the day and wake up at night to be active, eat, etc., the animals fed a high fat diet were eating at all hours of the night and day, even though they were eating roughly the same number of calories as their control counterparts. In order to determine if this aberrant feeding schedule was contributing to the disease, the animals were restricted to feeding during a specific time window during the night, matching when mice should normally be eating rather than letting them eat whenever they wanted (ad libitum). Surprisingly, the high-fat diet no longer resulted in mice with metabolic syndrome when they were restricted to a specific feeding time interval during the night. Even though these mice were allowed to eat as much as they wanted, those put on the time restricted feeding schedule did not gain weight, become diabetic, develop cardiovascular disease or anything of the like [9]. Similar experiments have been performed with humans and it has been demonstrated that when a pre-diabetic human only eats during the circadian time that they should be eating, they have significantly improved health, despite how many calories they eat or even what they eat [7]. This set of findings shows how incredibly important your circadian rhythm is for helping maintain health and normalcy in your body. By disrupting even a seemingly small aspect of one’s circadian rhythm, there could be catastrophic health consequences.
While this example of how disrupting the circadian clock contributes to severe health deficits is fascinating, it is certainly not the only example. The circadian clock is highly intertwined with the health and maintenance of the body. For many diseases, it is still unclear if the disease itself causes disruptions in the circadian rhythm or if circadian disruptions contribute to the progression of the disease. For most diseases the answer is probably both, but we do know one thing: the more scientists uncover the mechanisms driving disease onset and progression, the more we discover just how integral our circadian health is for our overall health and happiness. Perhaps then, the relatively recent increase we have seen in so many diseases, from diabetes to Alzheimer’s to psychiatric disorders, can be attributed to the growing disconnect between our bodies’ natural circadian rhythms and our behavior. For billions of years, life was dependent on the cycle of the sun, but now, in a relatively miniscule amount of time, humans have figured out how to gain some independence from the sun. You could hop in your car at 2:00 am and drive to the nearest Taco Bell, stare into their brightly lit screen as you order, and then binge on some delicious high fat, high carb food from their “fourth meal” menu. Talk about a confused circadian rhythm!
1. Stolper, D. A., & Keller, C. B. (2018). A record of deep-ocean dissolved O2 from the oxidation state of iron in submarine basalts. Nature, 553(7688), 323-327. doi:10.1038/nature25009
2. Masri, S., & Sassone-Corsi, P. (2018). The emerging link between cancer, metabolism, and circadian rhythms. Nature Medicine, 24(12), 1795-1803. doi:10.1038/s41591-018-0271-8
5. Baron, K. G., & Reid, K. J. (2014). Circadian misalignment and health. International Review of Psychiatry, 26(2), 139-154. doi:10.3109/09540261.2014.911149
8. Longo, V., & Mattson, M. (2014). Fasting: Molecular Mechanisms and Clinical Applications. Cell Metabolism, 19(2), 181-192. doi:10.1016/j.cmet.2013.12.008
3. Hirano, A., Yumimoto, K., Tsunematsu, R., Matsumoto, M., Oyama, M., Kozuka-Hata, H., . . . Fukada, Y. (2013). FBXL21 Regulates Oscillation of the Circadian Clock through Ubiquitination and Stabilization of Cryptochromes. Cell, 152(5), 1106-1118. doi:10.1016/j.cell.2013.01.054
6. Sharma, A., Lee, S., Kim, H., Yoon, H., Ha, S., & Kang, S. U. (2020). Molecular Crosstalk Between Circadian Rhythmicity and the Development of Neurodegenerative Disorders. Frontiers in Neuroscience, 14. doi:10.3389/fnins.2020.00844
7. Sutton, E. F., Beyl, R., Early, K. S., Cefalu, W. T., Ravussin, E., & Peterson, C. M. (2018). Early Time-Restricted Feeding Improves Insulin Sensitivity, Blood Pressure, and Oxidative Stress Even without Weight Loss in Men with Prediabetes. Cell Metabolism, 27(6). doi:10.1016/j.cmet.2018.04.010
9. Hatori, M., Vollmers, C., Zarrinpar, A., Ditacchio, L., Bushong, E., Gill, S., . . . Panda, S. (2012). Time-Restricted Feeding without Reducing Caloric Intake Prevents Metabolic Diseases in Mice Fed a High-Fat Diet. Cell Metabolism, 15(6), 848-860. doi:10.1016/j.cmet.2012.04.019
Patke, A., Young, M. W., & Axelrod, S. (2019). Molecular mechanisms and physiological importance of circadian rhythms. Nature Reviews Molecular Cell Biology, 21(2), 67-84. doi:10.1038/s41580-019-0179-2

Pingback: The 2020 Time Vortex and other Tales of Perception | NeuWrite San Diego
Pingback: Magnetoreception – a Quantum Sixth Sense | NeuWrite San Diego