 March
29
March
29
Tags
The animals snakes have nightmares about
The Box Jellyfish has venom so powerful that its human victims go straight into shock. The bite of a king cobra can result in the death of a healthy human adult within fifteen minutes. One drop of venom from a cone snail is capable of killing more than twenty humans. We live in fear of the world’s most venomous creatures, animals capable of delivering toxic cocktails of neurotoxins that can result in coma, paralysis, convulsions, and even death. These animals no doubt deserve our fear; however, there are many out there who are not so intimidated by these dangerous creatures, and even take the opportunity to challenge them. Many have developed immunity to some of the most dangerous toxins known to humankind.
Honey Badger Don’t Care
In early 2011, a YouTube video went viral, which featured comical commentary dubbed over National Geographic footage of a wild honey badger winning a fight with (and subsequently eating) a king cobra. Since then, nearly 2 million people have seen how the legendary “Honey Badger Don’t Care”. Not only have honey badgers been observed to survive bites from various venomous snakes, but 25% their diet consists of these animals. In 2015, a research group released a study detailing just how honey badgers are able to survive the venom from these dangerous snakes. Before we get into the details, let’s first review how venoms often wreak their toxic havoc.
Venom Toxicology
Venom is a fluid mixture of chemical weapons, mostly proteins, that have evolved as defense mechanisms in many creatures. Venom contains both neurotoxins, which affect the nervous system, as well as hemotoxins, which affect the blood. Here, we will focus on neurotoxins.
In the nervous system, neurons communicate with each other by sending various types of chemical messengers, known as neurotransmitters, across a small gap, where they bind to receptors on adjacent neurons. Molecules which are structurally similar to these neurotransmitters, including some neurotoxins, can also bind to these receptors. Such neurotoxins can have agonistic or antagonistic effects. Agonistic neurotoxins bind to these receptors (sometimes irreversibly) and activate them the same way that normal neurotransmitters would. These excitatory toxins can overstimulate neurons, and result in muscle spasms and rapid heart rate. Antagonistic neurotoxins bind these receptors and block them, inhibiting normal neural communication (see Jones’ piece on Botox!). This inhibition can lead to muscle paralysis, low heart rate, and respiratory failure.

In the case of many snake venoms, neurotoxins target a type of acetylcholine receptor (AChR), which facilitates communication from nerve cells to muscles. These neurotoxins bind irreversibly to the AChRs, effectively blocking them and preventing communication from the nerve cells to the muscles. This results in rapid paralysis, respiratory failure, and ultimately, death. It turns out that in honey badgers, hedgehogs, pigs, and mongooses, the neurotoxin binding site of the AChR has acquired subtle mutations which prevent toxin binding, while still allowing acetylcholine to bind (Drabeck, Dean, and Jansa, 2015). Consequently, these animals can survive a bite from a venomous snake, and can even afford to take on this dangerous creature.
In the image below, we see a king cobra facing its only natural predator: a mongoose.

“The venom of the viper is not a poison to the viper itself” – Fontana
Interestingly, these types of mutations which allow some animals to be immune to snake venom have also allowed some types of snakes to be immune to their own. Felice Fontana, an 18th century Italian physicist, conducted pioneering work on the resistance of vipers to their own venom. He injected the venom from one viper into another, as well as from one viper into itself, and did not see any signs that the venom was producing harmful effects. These results led to Fontana’s well-known aphorism: “The venom of the viper is not a poison to the viper itself”. This was the first experimental evidence for natural resistance to snake venom.
Vipers aren’t the only types of snakes that exhibit resistance to their own venom. For example, the AChR in Cobras shows the same binding resistance to venom neurotoxins as seen in the animals mentioned earlier (honey badgers, hedgehogs, pigs, and mongooses). Rattlesnakes also have some natural immunity to their venom; however, the self-immunity of rattlesnakes is due to an antivenom factor in their blood plasma, rather than a mutation in the neurotoxin binding site of the AChR. Further, venom immunity in snakes is not restricted to within a species. Following the work of Fontana, Guyon (1861) injected viper venom into other snake species and observed that other types of snakes also exhibited immunity to the venom. Finally, it should be noted that snakes aren’t completely immune to venom. The reaction (or lack thereof) that a snake has to an injection of venom depends on the type of snake, the type of venom, and the amount of venom injected. Regardless, the ability to survive such powerful toxins is an impressive feat.
So, why study venom?
In 2017, researchers found a particular peptide in the blood of opposums which they claimed neutralized the effect of snake venom in mice. When they mixed a lethal dose of the snake venom with the peptide prior to injecting the mixture into mice, they found no evidence of the mice being sick. These results garnered media attention, leading some to wonder if studies like this could hold the key to developing a universal antivenom. While it’s unlikely that a single peptide could neutralize the hundreds of different harmful compounds within a venom, findings such as these provide us with a better understanding of how our immune systems function, as well as better tools to treat venom poisoning.
Studies on toxins and venoms have opened several exciting avenues for fighting human disease. For example, the studies on snake venom immunity helped scientists better define the AChR at the molecular level. This receptor plays a big role in diseases such as myasthenia gravis and congenital myasthenic syndrome. Further, studies on snake venom have played a huge role in developing our understanding of blood coagulation and basic immune functioning (Zhang, 2015), which have implications for a wide range of medical conditions. As seen in the figure from Zhang (2015) below, the toxins in venom offer “an invaluable source of natural products for drug discovery”.
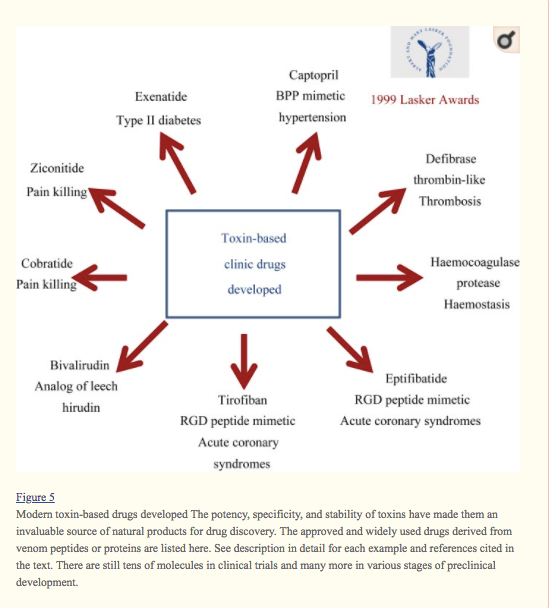
Despite the dangers venomous animals pose to our health (and even our lives), studying these venoms and how they react with the immune systems of various animals ultimately helps us better understand ourselves.
References
Drabeck DH, Dean AM, Jansa SA. (2015). Why the honey badger don’t care: Convergent evolution of venom-targeted nicotinic acetylcholine receptors in mammals that survive venomous snake bites. Toxicon 1;99:68-72. doi: 10.1016/j.toxicon.2015.03.007
Fontana F., Richerche Fisiche Sopra il veleno Della Vipera. Lucca J, Guisti, Treatise on the venom of the viper, Murray, London, England, 1787.
Matthew L. Holding, Danielle H. Drabeck, Sharon A. Jansa, H. Lisle Gibbs; Venom Resistance as a Model for Understanding the Molecular Basis of Complex Coevolutionary Adaptations, Integrative and Comparative Biology, Volume 56, Issue 5, 1 November 2016, Pages 1032–1043, https://doi.org/10.1093/icb/icw082
Striaght R, Glenn JL, Snyder CC. (1976). Antivenom activity of rattlesnake blood plasma. Nature 261, pages 259–260. doi:10.1038/261259a0
Guyon J. Le Venin de Serpents Exerce-t-il sur Eux-mêmes l’Action Qu’il Exerce sur D’autres Animaux? In: Phisalix M., editor. Animaux Venimeux et Venins: La Fonction Venimeuse chez tous les Animaux; les Appareils Venimeux; les Venins et leurs Propriétés; les Fonctions et Usages des Venins; L’envenimation et son Traitement. Volume 2. Masson & Cie.; Paris, France: 1861. p. 744.
Komives CF, Sanchez EE, Rathore AS, White B, Suntravat M, Balderrama M, Cifelli A, Joshi V. (2017). Opossum peptide that can neutralize rattlesnake venom is expressed in Escherichia coli. Biotechnol Prog 33(1): 81-86. doi: 10.1002/btpr.2386
Zhang Y. (2015). Why do we study animal toxins?. Dongwuxue Yanjiu 36(4): 183-222. doi: 10.13918/j.issn.2095-8137.2015.4.183

You must be logged in to post a comment.