 May
31
May
31
The Rise and Fall and Rise of Genetic Memory
We are all products of our past, for better or for worse. At first glance, such a statement seems so obvious it hardly bears mentioning; our earlier experiences, both our successes and our failures, shape our current behavior. But dig just a bit deeper, and it becomes far murkier. What can you call your past? Is your history just your own experiences, or does it include your heritage too?
An Origin in Time Immemorial
Today, most people tend to define themselves independently from their ancestors. No matter how clear such a definition may seem today, this consensus is less than 50 years old. Instead, for most of history, the delineation between a person and their ancestors was far less stark. This idea featured most prominently in Buddhism and Hinduism, where it is believed that a being’s actions in their past life determined the conditions of their current life, and the most enlightened could allegedly remember the events of their past lives. The classical Greeks had their own parallel in metempsychosis (“transmigration of the soul”), and in the Norse sagas, the wounds of some heroes’ ancestors would appear on their own bodies.
These ideas actually held purchase in the first years of psychology. William James, the father of American psychology, was notorious for his strong belief in reincarnation, and used his significant influence to advance its study in psychology [1]. Carl Jung also believed in a continuity of personality over multiple lives, which was key to his initial development of his collective unconscious concept. These ideas also led to the creation of past life regression, where hypnosis can supposedly allow patients to recall their past lives. However, absolutely no credible evidence for its efficacy has ever been found, and most, if not all, psychologists now consider these ideas “certainly discredited” [2].
These ideas did have a second incarnation, though, in Richard Semon’s concept of “genetic memory” [3]. Genetic memories were proposed as memories that could be stored and transferred to other individuals genetically in the absence of their own experience. On its face, genetic memories made far more sense than memories of past lives. After all, evolution provides a vehicle for past individual experiences (in this case, adaptation and reproduction) to affect the biology of the following generations, transmitted through the genes. If such processes affected the rest of the body, why not the mind as well? The genetic memory concept gathered influence in some corners of psychology, especially to explain instincts, which behaviorism, the leading theory at the time, could not easily address.
The Birth, Brief Life, and Death of Genetic Memory
Though the genetic memory concept was first developed in 1921, it could only be tested after major advances in behavioral and biochemical techniques in the 1950s. James McConnell, a then-rising star in psychology at the University of Michigan, published the first paper testing the concept using flatworms. McConnell took advantage of flatworms’ inherent capability to regenerate and their readiness to cannibalize each other to test whether memories could be transferred between individuals. First, McConnell would train a flatworm to avoid a flash of light by shocking it shortly after a flash, which causes it to immediately turn. Then, he would either cut off the worm’s head or feed them to other worms, and the next worm would apparently remember the last worm’s training, turning after the flash like the first worm [4]. A follow-up study then identified RNA as the substance carrying the memory from worm to worm by isolating RNA from trained flatworms and injecting it into untrained flatworms, who then appeared to learn the behavior and turn as well [5].
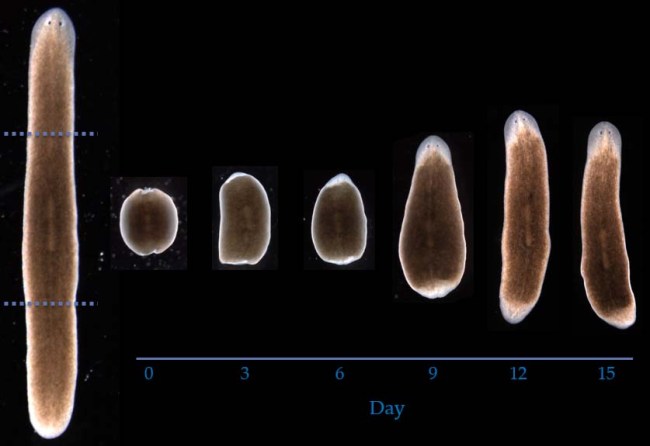 The flatworm has a remarkable ability to regenerate from any part of its body when completely severed (Lobo Lab; University of Maryland, Baltimore County).
The flatworm has a remarkable ability to regenerate from any part of its body when completely severed (Lobo Lab; University of Maryland, Baltimore County).
These results quickly took the psychology community by storm. Interest was even further heightened after Georges Unger, a professor at Baylor, trained rats to fear darkness, and then transferred extracts from their brains to other rats, who then seemed to remember the training [6]. He isolated this effect to a peptide in the extract, which he then synthesized and christened ‘scotophobin’, claiming it directly encoded that specific fear information in the rats’ brains [7].
At the time, it appeared as if McConnell, Unger, and their collaborators were on the verge of birthing a new subfield. However, cracks soon developed in this nascent paradigm’s foundation. The first dissent was authored by the Nobel Laureate Melvin Calvin, who could not replicate McConnell’s results in flatworms. Soon after, dozens of labs reported an inability to replicate McConnell’s findings. Finally, McConnell himself reported that he had failed to clean the chambers between trials, and that the effect disappeared with clean chambers [8]. The worms just followed the trails of the worms they were fed. As the final nail in the coffin, another group could not replicate most behavioral effects of scotophobin, and the few they did observe occurred in a different rat fear memory test too, showing this molecule simply caused anxiety, instead of transferring a memory [9].
The paradigm of genetic memory was stillborn, its fall to earth even quicker than its meteoric rise. McConnell and Ungar were written out of history; these once-landmark papers have disappeared from all contemporary histories of memory: their roles filled by Kandel and Carew, the flatworm replaced by the sea slug, and connections between brain cells became the favored medium for memory storage over genes. James McConnell was professionally ostracized and turned to popular science writing instead, while Georges Ungar died in 1977, on the cusp of scotophobin’s failure. With no one to pick up their torch, their work went dormant and appeared to be consigned to the dustbins of scientific history.
An Epigenetic Reincarnation
Until almost four decades later. In 2014, Kerry Ressler, a fear memory researcher at Emory (now at Harvard), reported a strange, provocative result. When he trained mice to fear cherry blossom odor (acetophenone), he found that the mice’s children and grandchildren also feared the odor, despite having never been trained themselves, and unlike Ungar’s work, the effect was specific to this cherry blossom odor alone, instead of generalized anxiety [10]. Even when they used in-vitro fertilization or separated the children from their parents at birth, the effect persisted. Further, the cells that detected this odor appeared more abundant than the cells in mice descended from untrained ancestors. They had turned a learned fear into an instinctive one, seemingly by hardwiring it into the structure of the nervous system.
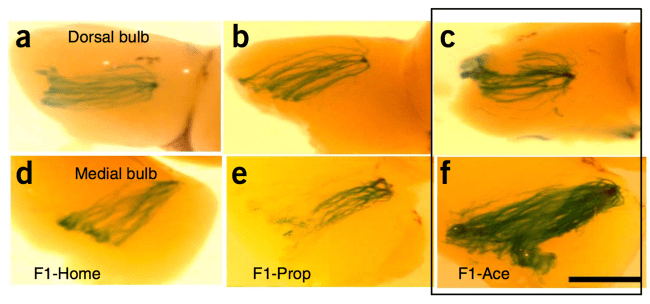
Cells in the olfactory bulb bearing the receptor for cherry blossom odor (Ace) are marked in blue; c and f (outlined in black) are from mice whose ancestors were trained using the odor, while the others are not. Reproduced from [10].
Searching for a mechanism, he examined the sperm of the trained mice and their offspring. Interestingly, both had a very specific, limited epigenetic trace (i.e. changes to markings on genes separate from the genes themselves) that would drastically increase the amount of cherry blossom odor receptor their cells would produce. This finding was even more bewildering. So bewildering, in fact, that he published a separate paper outlining all the questions these results raised and all the assumptions they violated [11]. For example, how did a pattern of nervous system activity get transferred to sperm, and how did an epigenetic signal, which is usually temporary, become heritable? To this day, these questions remain unanswered, though they are currently under intense study.
That same year, David Glanzman, a UCLA neuroscientist, made a striking, related discovery. Using sea slugs trained to recoil from a stimulus, he was able to induce amnesia using chelerythrine, a drug that can reset the connections in the sea slug’s nervous system to their state before learning. After the slugs seemed to lose their memory after chelythrine treatment, they would quickly and spontaneously recover their training, even though it should have permanently erased their memory [12].
When he investigated a possible mechanism, he found the same thing Ressler did. Maintenance of an epigenetic signal was crucial to maintain the memory, and removal of this signal removed the memory, whether or not the connections were maintained [13]. Just last month, they also found RNA from trained sea slugs could induce a trained response in untrained sea slugs as well, like in McConnell’s original paradigm, but only if the epigenetics were maintained [14].
The history of genetic memory is a complex and confusing one, full of research programs lurching forward in fits and starts, usually down blind alleys, and often abandoned. It remains to be seen whether or not its latest epigenetic incarnation will face the same fate as its predecessors, or if it will defy history. Only one thing is for certain: its journey will surely be interesting to follow.
References
- Sech Jr., A., de Freitas Araujo, S., & Moreira-Almeida, A. (2012). William James and psychical research: toward a radical science of mind. Hist. Psychology, 24(1), 62-78. doi: 10.1177/0957154X12450138.
- Norcross, J.C., Koocher, G.P., & Garofalo, A. (2006). Discredited psychological treatments and tests: A Delphi poll. Prof. Psychol.: Res. Pract., 37(5), 515-522.
- Semon, R. (1921). The Mneme. London: Allen and Unwin.
- McConnell, J.V. (1962). Memory transfer through cannibalism in planarians. J Neuropsychiatry, 3 Suppl 1, S42-46.
- Jacobson, A.L., Fried, C., & Horowitz, S.D. (1966). Planarians and memory. Nature, 209(5053), 599-601.
- Ungar, G. & Oceguera-Navarro, C. (1965). Transfer of habituation by material extracted from brain. Nature, 207(994), 301-302.
- Ungar, G., Desiderio, D.M., & Parr, W. (1970). Isolation, identification, and synthesis of a specific-behaviour-inducing brain peptide. Nature, 238(5361), 198-202.
- Travis, G.D.L. (1981). Replicating replication? Aspects of the social construction of learning in planarian worms. Soc. Stud. Sci., 11(1), 11-32. doi: 10.1177/030631278101100102.
- Misslin, R., Ropartz, P., Ungerer, A., & Mandel, P. (1978). Non-reproducibility of the behavioural effects induced by scotophobin. Behav. Processes, 3(1), 45-56. doi: 10.1016/0376-6357(78)90029-3.
- Dias, B.G. & Ressler, K.J. (2014). Parental olfactory experience influences behavior and neural structure in subsequent generations. Nat. Neurosci., 17(1), 89-96. doi: 10.1038/nn.3594.
- Dias, B.G. & Ressler, K.J. (2014b). Experimental evidence needed to demonstrate inter- and trans-generational effects of ancestral experiences in mammals. Bioessays, 36(10), 919-923. doi: 10.1002/bies.201400105.
- Chen, S., Cai, D., Pearce, K., Sun, P.Y.-W., Roberts, A.C., & Glanzman, D.L. (2014). Reinstatement of long-term memory following erasure of its behavioral and synaptic expression in Aplysia. eLife, 3, e03896. doi: 10.7554/eLife.03896.
- Pearce, K., Cai, D., Roberts, A.C., & Glanzman, D.L. (2017). Role of protein synthesis and DNA methylation in the consolidation and maintenance of long-term memory in Aplysia. eLife, 6, e18299. doi: 10.7554/eLife.18299.
- Bédécarrats, A., Chen, S., Pearce, K., Cai, D., & Glanzman D.L. (2018). RNA from trained Aplysia can induce an epigenetic engram for long-term sensitization in untrained Aplysia. eNeuro, 5(3), ENEURO.0038-18.2018. doi: 10.1523/ENEURO.0038-18.2018.

You must be logged in to post a comment.