 April
25
April
25
Tags
Immune to pain: new insights into chronic pain treatment
Jo Cameron, a Scottish woman in her mid-60s, was seemingly happy and healthy other than a problem with her hip. Now and then, it would give way and prevent her from walking straight. She had brought it up to her doctor, but because she wasn’t in pain, the issue was dismissed. It wasn’t until a few years later while taking an x-ray when a doctor realized that the joint in her hip was highly deteriorated. Already surprised that she hadn’t been in intense discomfort or pain this whole time, the doctor was even more taken aback when Jo turned down pain medication for the surgery that would be required to repair her hip. She simply took some tylenol the couple of days following the procedure. While there, doctors also noticed severe osteoarthritis in her thumbs, which would require an extremely painful surgery on both of her hands. Once again, she turned down pain medication, and seemed perfectly fine with a couple of tylenol after the surgery. Shocked, a consultant referred Jo to pain specialists at University College London (UCL), where a team of researchers would dig into her past and her genetics to determine the cause of her apparent immunity to pain. [1,2]
A Far-Out Condition
At UCL, a team of researchers led by Drs. Devjit Srivastava and James Cox [3] learned about Jo’s history of painless injuries. She never requested or seemed to require pain medication for things like varicose vein treatment or dental procedures, and she once had a cut stitched up without any analgesic. She described childbirth as a “tickle”, and could eat Scotch bonnet chili peppers (which are 32-50 times hotter than jalapeno peppers!) without the slightest discomfort — only the sensation of a “warm glow”. Interestingly, she also reported never feeling anxiety, fear, or major sadness. Although it may sound wonderful to not have to experience any pain, pain serves a purpose: to alert us when a part of our body is being damaged (see Catie’s previous post to learn about how pain is essential for our survival!). For example, Jo also reported often leaning against the oven and burning herself, only realizing what was happening when she smelled her skin burning.
To figure out why this was happening, Drs. Srivastava and Cox and their team of scientists took a sample of Jo’s DNA and looked for any genetic abnormalities that might explain her pain insensitivity. They found two mutations: a single nucleotide polymorphism (SNP: pronounced “snip”) in a gene called FAAH, and a microdeletion in a previously unknown gene which they called FAAH-OUT.
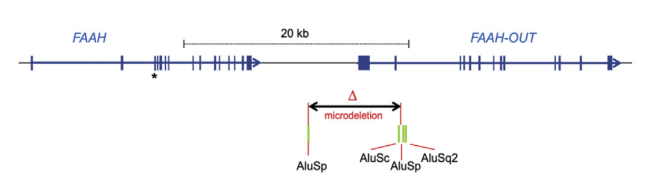
Taken from [3]: “Genomic map showing FAAH, FAAH-OUT, and microdeletion”
The second mutation the UCL researchers found in Jo’s DNA had never been found before– a deletion of a small part of DNA in a previously unknown gene nearby FAAH, which they named FAAH-OUT. They hypothesized that FAAH-OUT acts as a kind of “volume control” for FAAH. In other words, they think that the FAAH-OUT microdeletion further disrupts the function of FAAH on top of the SNP, resulting in even less breakdown of anandamide.

Taken from [3]: Circulating anandamide (AEA) levels from the patient and four controls. Controls A and B don’t have the SNP; Controls C and D do.
The Endocannabinoid System in Pain & Anxiety
Jo is not the only known case of a person unable to feel pain, a condition sometimes called congenital insensitivity to pain (CIP). While extremely rare, many of these cases are known to be due to a mutation in the gene SCN9A, which encodes an ion channel important for electrical signaling in pain neurons of spinal nerves. However, Jo is the only known case of someone with CIP due to the co-inheritance of the FAAH SNP and FAAH-OUT microdeletion.
This may explain why Jo also reports feeling no anxiety, fear, or depression, which isn’t something typically seen in people with pain insensitivity. Anandamide is an endocannabinoid, meaning that it binds to the same receptors as other exogenous cannabinoids such as THC and CBD from the cannabis plant (side note: cannabinoid receptors were found and named for their sensitivity to these cannabinoids, and anandamide was later discovered through further research into these receptors as a naturally produced cannabinoid – hence the name endocannabinoid! [5]). Endocannabinoids, including anandamide, play a role in pain perception, fear, anxiety, and depression, among other things [6,7]. Thus, the endocannabinoid system is an attractive target for treating chronic disorders of these symptoms.
New Insights Into Chronic Pain Treatment
Given our growing knowledge about the functions of anandamide, scientists have tried to target the FAAH gene as a potential way to treat chronic pain, anxiety, and depression. Recent clinical trials have aimed to inhibit FAAH in order to increase anandamide levels in the body. Unfortunately, these trials have been unsuccessful so far [8,9]. However, Jo Cameron’s case provides new insights about the possibilities of developing FAAH-related analgesia through targeting of FAAH-OUT instead. With the ever-growing opioid epidemic, we are in major need of novel, safe, and effective treatments for the millions of people living with chronic pain. Targeting of FAAH-OUT may be a promising next step for pain researchers to pursue.
References
- Habib, Okorokov, Hill et al. (2019). Microdeletion in a FAAH Pseudogene Identified in a Patient with High Anandamide Concentrations and Pain Insensitivity. Br J Anaesth pii: S0007-0912(19)30138-2. doi: 10.1016/j.bja.2019.02.019
- Cajanus, Holmström, Wessman, Anttila, Kaunisto, Kalso (2016). Effect of endocannabinoid degradation on pain: role of FAAH polymorphisms in experimental and postoperative pain in women treated for breast cancer. Pain. 157(2):361-9. doi: 10.1097/j.pain.0000000000000398.
- Devane, Hanus, Breuer, Pertwee, Stevenson, Griffin, Gibson, Mandelbaum, Etinger, Mechoulam (1992). Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258(5090): 1946-1949. DOI: 10.1126/science.1470919
- Woodhams SG, Sagar DR, Burston JJ, Chapman V. The role of the endocannabinoid system in pain. Handb Exp Pharmacol 2015; 227: 119e43
- Mechoulam R, Parker LA. The endocannabinoid system and the brain. Annu Rev Psychol 2013; 64: 21e47
- Huggins JP, Smart TS, Langman S, Taylor L, Young T. An efficient randomised, placebo-controlled clinical trial with the irreversible fatty acid amide hydrolase-1 inhibitor PF04457845, which modulates endocannabinoids but fails to induce effective analgesia in patients with pain due to osteoarthritis of the knee. Pain 2012; 153: 1837e46
- Kerbrat A, Ferre JC, Fillatre P, et al. Acute neurologic disorder from an inhibitor of fatty acid amide hydrolase. N Engl J Med 2016; 375: 1717e25


You must be logged in to post a comment.