 September
03
September
03
Tags
Microglia as Architects and Designers for Your New Brain
[En español] While neurons tend to get the spotlight in the brain, there’re tons of other cell types working in the background to support brain health and function. Microglia are one such cell type, often described as the immune cells of the brain, patrolling and gobbling things up like white blood cells do in the rest of your body. Researchers have discovered that they also play an important role in shaping how neurons function and communicate, for example by sensing neuronal activity and working to prune away unwanted or unused synapses. These functions make microglia an important player in shaping the brain as it encounters new experiences, disease, and injury.
But microglia also play key roles in how the brain first sets itself up, before you’re even born. These cells are born very early on and travel into the brain to become the first glial cells in the brain. This means that microglia are present before many neuronal cell types have even formed and thus are in a perfect position to influence the assembly and maturation of the brain (1,2). And as cells that can sense activity levels in both the immune system and brain, microglia could be a key link for explaining why infections and inflammation during pregnancy are associated with neurodevelopmental disorders like autism spectrum disorder (ASD).
To tackle this chain of connections, let’s follow microglia through some of the major steps in brain development:
First stop: neurogenesis
Neuronal progenitors are stem cells that produce neurons to populate the brain during early development; this process is called neurogenesis. The progenitors that produce all the excitatory neurons in the cortex—the wrinkly outer portion of the brain—are found in regions buried deep in the center of the brain, the VZ and SVZ, as shown in Figure 1. Neurogenesis must be tightly controlled so that the correct numbers and types of cells are produced at the right times. Since this is one of the first stages of brain development, abnormalities here—for example, too many or too few neurons produced—can disrupt everything that happens next, such as arranging cells in layers, forming synapses, and setting up brain circuits. So how is neurogenesis controlled? Microglia act as one way to put the brakes on neuronal progenitors.
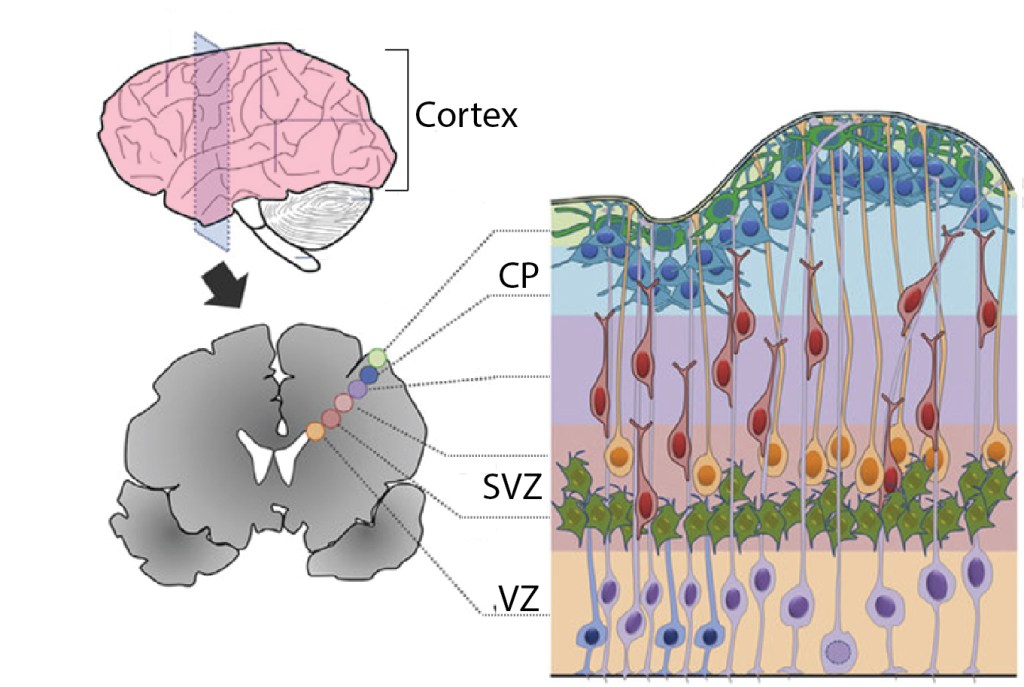
Microglia are dispersed relatively homogenously throughout the mature brain. But in the early brain, microglia first congregate at key ‘focal points’ where they influence neuronal development. At first, microglia largely avoid the cortical plate (Fig. 2A). Then, as neurogenesis kicks off and the layers grow thicker, microglia pour into the VZ and SVZ to mingle with the progenitor cells (Fig. 2C-D). They gradually enter an ‘activated’ state and begin to phagocytose—eat and digest—-the progenitor cells! Just by manipulating the microglia, for example by blocking them from entering the activated state, researchers could change the number of progenitors, thus potentially changing the number and types of neurons produced (3). Like a manager at a construction site, microglia make sure that the right amount of building materials are at hand to properly put together the cortex.

Trailguides for wandering neurons and axons
The brain has a super complex structure, with lots of cell types mixed together, organized layers, and intricate wiring between different brain regions. Several steps are required after neurogenesis to achieve this complex organization. For example, many cell types can’t stay where they’re born—inhibitory interneurons are another key type of cortical neuron, but they’re actually born far away from the cortex. These interneurons embark on an epic journey to find the cortex and disperse among the regions and layers. One of the main routes they take is the SVZ, which as we saw earlier is also a hotspot where microglia gather. When researchers removed microglia, interneurons arrived in the cortex abnormally early and were abnormally distributed. Though it’s not clear exactly how microglia shape interneuron migration, they’re now being seen as a kind of ‘guidepost cell’, like helpful park rangers who scope out the trail first and leave behind trail markers for other traveling cells.
Microglia may also play an important role in setting up the connections between brain regions. Neurons that produce the neurotransmitter dopamine send axons into the striatum to form part of a circuit that is key to functions such as movement, motivation, and decision-making. During development, these axons must grow into the striatum, and during this stage microglia start to gather around their edges. Closer examination showed that these microglia were actually eating up parts of the dopaminergic axons, like pruning shears keeping growing branches in line. Again, the phagocytic immune-cell powers of microglia have been harnessed to shepherd wandering axons to specific destinations, shaping the physical structure of the brain. If microglia are removed only during development, these dopaminergic axons overgrow and spread too far into the striatum (Fig. 3), causing lasting changes to the circuit, even if microglia return later on (4).

Brain development tunes into the outside world
One of the most important functions of the brain is to sense what’s happening outside of it, whether that be the body or the world outside the body. This takes on a whole new meaning during development, when the surrounding environment is another body. Yet infections during pregnancy and the subsequent immune response they elicit in a mother’s body have been associated with increased risk for ASD and schizophrenia, which suggests that the developing brain does respond to the immune environment of the womb. There’s still much unknown about how maternal immune activation (MIA) contributes to brain dysfunction, but microglia could be a key piece of this puzzle. Microglia sense immune activation, which could include molecules or inflammation from the mother’s body, and enter an activated state. Now we know that microglia change the way they shape brain development after becoming activated. For example, after inducing MIA in mice, researchers found that the extension of dopamine axons was reduced—suggesting that they were over-pruned by overactive microglia (Fig. 3). Then after birth, these mice showed less dopamine innervation in the dorsal striatum—associated with things like movement and habit formation—and extra dopamine innervation in the ventral striatum—which is important for parsing pleasure and punishment (4). Thus early changes in microglia activity—like those caused by inflammation in the mother—could create imbalances in key brain circuits and contribute to dysfunction later in life.
These discoveries on microglial roles specific to early development also illustrate a key point on why studying brain development is so cool and so important: there are cell types and functions that only exist during development. As the brain matures these cells take on other roles—microglia act as guideposts during early development, but then disperse and take on immune functions. If we only look at the fully-formed brain, it’s difficult to see the common developmental links between different brain structures—the fact that perturbing one cell type (microglia) can have downstream effects on excitatory cortical neurons, inhibitory cortical neurons, dopamine neurons, and maybe even other cells in other brain regions (5). If we understand development, maybe we could finally connect the dots on complex brain disorders that have been so difficult to study and treat.
References
- Squarzoni P, Thion MS, Garel S. Neuronal and microglial regulators of cortical wiring: Usual and novel guideposts. Front Neurosci 2015;9:248. https://doi.org/10.3389/fnins.2015.00248.
- Thion MS, Garel S. On place and time: microglia in embryonic and perinatal brain development. Curr Opin Neurobiol 2017:121–30. https://doi.org/10.1016/j.conb.2017.10.004.
- Cunningham CL, Martínez-Cerdeño V, Noctor SC. Microglia regulate the number of neural precursor cells in the developing cerebral cortex. J Neurosci 2013;33:4216–33. https://doi.org/10.1523/JNEUROSCI.3441-12.2013.
- Squarzoni P, Oller G, Hoeffel G, Pont-Lezica L, Rostaing P, Low D, et al. Microglia Modulate Wiring of the Embryonic Forebrain. Cell Rep 2014;8:1271–9. https://doi.org/10.1016/j.celrep.2014.07.042.
- Fishell, G. Making up Your Mind: Interneurons in Development and Disease. SFARI 2016. https://www.sfari.org/event/making-up-your-mind-interneurons-in-development-and-disease/
- Romero DM, Bahi-Buisson N, Francis F. Genetics and mechanisms leading to human cortical malformations. Semin Cell Dev Biol. 2018;76:33-75. doi:10.1016/j.semcdb.2017.09.031

Pingback: Nyheder2020september03 - bionyt.dk
Pingback: Your brain is plastic! | NeuWrite San Diego