 January
09
January
09
Tags
Can we inherit family trauma?
The epigenetics behind “generational trauma”
The 23 and Me craze has officially reached my family. Both of my parents were born in Poland so the results weren’t a complete surprise, but as we watched the site track generations of family history from a single spit sample, new questions came up. When my sisters and I found ourselves around a table with my parents and some time to spare, we decided to ask more about our family tree. It started with the names and places we knew, but the tree quickly grew, and with each new branch came a series of characters and stories that were hard to believe. We took notes. I learned of big families on small farms. Teens sent on ships towards foreign lands to find work and send back money. Parents lost children; children lost parents. Each life personally affected by two world wars. And there was one banishment to Siberia thrown in for good measure. I left thinking not just about my own family, but about how all families carry these incredible stories of survival, through forms of struggle that many of us will not have to experience ourselves. And yet, emerging evidence for the idea of generational trauma suggests that recent generations have not come out unscathed.
In the everlasting nature vs. nurture debate, we rely on the fact that our genetic code is unaffected by our life experiences. Our behavior and health are certainly shaped by our lifestyles, but we can only pass these traits down to our kids through social and cultural means. Or so we thought. A new form of transgenerational inheritance has now been suggested, in which the effects of trauma and stress in a parent’s life can actually be passed on to children in ways that shape their physical, behavioral, and cognitive health. Many studies have already used this idea to explain health issues observed in descendants of Holocaust survivors, refugees, veterans, and those who have endured poverty, racism, and natural disasters [1-4]. But how could this work?
While your life experiences do not affect your genetic code (what those 23 and Me results are based on), they do affect your epigenetics. Surrounding your DNA is a complicated system of molecular tools that decide which genes will get turned on and off, when and where. If your genes are the ingredients list in a recipe, your epigenetics are the instructions for what to do with them. And the system responds to changes in the environment, so when a traumatic event occurs, your body will react and write new instructions. What we didn’t know was that those new instructions could then be passed on to your children. These epigenetic tools play a major role in guiding embryonic development, so small changes in the instructions can lead to a cascade of molecular events that shape the function of your brain, body, and behavior.
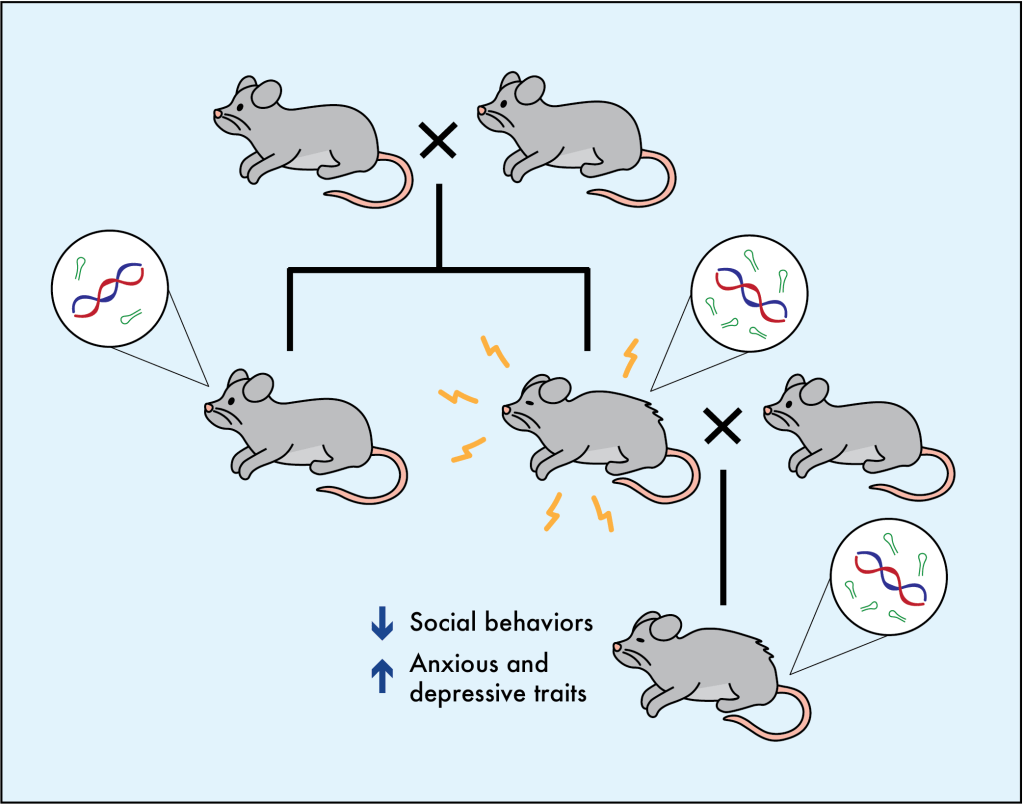
This has huge implications. But many scientists remain skeptical of the human studies, especially since it is so difficult to tease apart which traits are transmitted epigenetically vs. through the social dynamics that result from trauma. More reliable data comes from mouse studies, where these different factors can be more thoroughly controlled. In these experiments, mice are subjected to a stressful event such as a painful shock or parental separation, which induces a measurable change to their epigenetics [5-7]. For example, in male mice exposed to stress, their sperm show sudden changes in the levels of epigenetic molecules called microRNA, which work to block the expression of specific genes. Interestingly, offspring of these mice behave differently than other mice, showing more anxious and depressive traits, acting less social, and taking more risks. Furthermore, the male offspring of the stressed mice show the same epigenetic alterations in their sperm (e.g. more microRNA), even though they haven’t experienced the trauma themselves. Importantly, mice are not raised with their fathers, so these changes don’t happen through social interactions with stressed parents. To further prove these behaviors come from the epigenetic changes, scientists have also injected the microRNAs directly into embryos and see the same behavioral changes in those animals and their offspring, even when no traumatic event occurred. One study was actually able to show that these epigenetic changes lead to the genetic reprogramming of the hypothalamus, a brain area involved in regulating stress responses [7].
Can we relate these mouse experiments back to us? In an especially interesting study, a group looked at these microRNAs in both mice and men. They had the adult men complete the Adverse Childhood Experiences (ACE) survey, which asks how many forms of abuse or neglect were experienced during a person’s childhood. A high ACE score is strongly correlated with stress-associated behavioral and health problems, for that individual and their children. The researchers then compared the epigenetic profile of the men’s sperm to the sperm of stressed mice. Surprisingly, they found changes in the same microRNAs in both humans and mice, and the higher the men’s ACE score was, the more likely they were to match the epigenetic profile of the stressed mice [8].
So is there anything we can do about this? The behavioral effects of traumatic stress have been shown to get passed down through at least four generations [9] – but can we stop the cycle? Some studies do suggest potential ways to reverse the epigenetic changes, including good maternal health and nutrition during pregnancy. Though we still have a lot to learn about epigenetics and how it shapes our brain function, a better understanding may help us develop biological tools to selectively reprogram the system. But until we can target the molecular level directly, we can at least try to mitigate the effects of generational trauma. California has recently appointed Dr. Nadine Burke Harris as the state’s first Surgeon General, who is working to incorporate the ACE survey of childhood trauma into standard doctor’s visits, for both children and their parents to complete. This is done with the knowledge that early intervention through medical care, psychological support, and nurturing environments can significantly improve health outcomes. Knowing your own family history of trauma can be critical in this way (you can’t fix what you don’t know). And if our epigenetics are indeed capable of responding flexibly to our current circumstances, maybe treating the trauma will be enough to reach the molecular source. But perhaps most effectively and importantly, increasing awareness of the long-lasting damage that trauma can cause may help us prevent it from occurring in the first place, as best we can.
References:
- Yehuda R, Daskalakis NP, Bierer LM, Bader HM, Klengel T, Holsboer F, Binder EB. (2016) Holocaust Exposure Induced Intergenerational Effects on FKBP5 Methylation. Biological Psychiatry, 80:372–380.
- Costa DL, Yetter N, DeSomer H. (2018) Intergenerational transmission of paternal trauma among US Civil War ex-POWs. PNAS, 115(44): 11215–11220.
- Vågerö D, Pinger PR, Aronsson V, van den Berg GJ. (2018) Paternal grandfather’s access to food predicts all-cause and cancer mortality in grandsons. Nature Communications, 9: 5124.
- Aroke EN, Joseph PV, Roy A, Overstreet DS, Tollefsbol TO, Vance DE, Goodin BR. (2019) Could epigenetics help explain racial disparities in chronic pain? Journal of Pain Research, 12: 701–710.
- Dias BG and Ressler KJ. (2014) Parental olfactory experience influences behavior and neural structure in subsequent generations. Nature Neuroscience, 17(1): 89–96.
- Gapp K, Jawaid A, Sarkies P, Bohacek J, Pelczar P, Prados J, Farinelli L, Miska E, Mansuy IM. (2014) Implication of sperm RNAs in transgenerational inheritance of the effects of early trauma in mice. Nature Neuroscience, 17(5): 667–669.
- Rodgers AB, Morgan CP, Leu NA, Bale TL. (2015) Transgenerational epigenetic programming via sperm microRNA recapitulates effects of paternal stress. PNAS, 112(44): 13699–13704.
- Dickson DA, Paulus JK, Mensah V, Lem J, Saavedra-Rodriguez L, Gentry A, Pagidas K, Feig LA. (2018) Reduced levels of miRNAs 449 and 34 in sperm of mice and men exposed to early life stress. Translational Psychiatry, 8: 101.
- van Steenwyk G, Roszkowski M, Manuella F, Franklin TB, Mansuy IM. (2018) Transgenerational inheritance of behavioral and metabolic effects of paternal exposure to traumatic stress in early postnatal life: evidence in the 4th generation. Environmental Epigenetics, 4(2).
Cover Image by Marco Verch.
License: https://creativecommons.org/licenses/by/2.0/

You must be logged in to post a comment.