 May
25
May
25
Tags
Cranium-bound Ultrasound: A Novel Brain Stimulation Method
Over the last few years, the application of ultrasonic stimulation has been heading towards a rather intimate part of the human body: the brain. Researchers have engineered a way to transmit ultrasound through the skull to influence brain activity, remarkably without the necessity of brain surgery. While non-invasive brain stimulation has been around for quite some time, the nature of transcranial ultrasound (TUS) has opened doors for more promising applications. Unlike other brain-stimulation methods, TUS can be aimed with millimeter precision towards structures embedded deep in the brain and is free of physical sensations and sounds that might otherwise inform naive participants to a study’s experimental treatment conditions. This up-and-coming technology also carries the benefit of tapping into already existing ultrasound infrastructure in hospital and clinics. Most importantly, its long history of clinical use has led to the calculations necessary for its safe application on biological tissue such as skin. Finally, due to the mechanical nature of ultrasound, it’s implementation within a magnetic resonance imaging technique sets up the prospect of functional brain mapping.
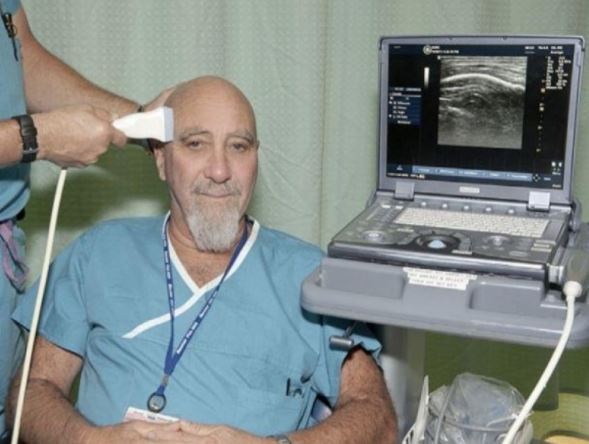
Stuart Hameroff at the University of Arizona led the first clinical study showing that transcranial ultrasound affects mood.
What it is
Ultrasound consists of mechanical vibrations induced by acoustic (sound) pressure. The reason we can’t hear ultrasound is because the frequency at which it occurs is far higher than the upper limit of human hearing. The intensity, or acoustic pressure parameter, at which ultrasound is applied results in various outcomes. For example, the entire human body has been imaged using low-intensity ultrasound whereas high-intensity ultrasound has been medically approved for heating and removing kidney stones. Ultrasound at a frequency of around 1 megahertz (MHz) has therapeutic applications, while frequencies between 1MHz-15MHz are useful for diagnostic imaging. An ultrasound technician delivers pulses between brief intermissions, monitoring the temperature changes in targeted tissue resulting from the ultrasound via a thermal index (TI), as well as the mechanical pressure itself via a mechanical index (MI).

Representative sound frequency ranges. Human audition is limited to the 20 to 20,000 Hz range.
Animal studies and development
Early studies of ultrasound on nervous tissue found that the activity of nerve fibers could be modulated by varying degrees of ultrasound intensity. For example, by aiming ultrasound at the brain of a live cat, scientists recorded subsequent suppression of electrical activity in the visual cortical areas [1]. Later, it was found that low intensity ultrasound elicited neuronal activity with millimeter precision in post-mortem brain preparations of rodent hippocampus, an area of the brain heavily implicated in the formation of new memories [2]. A more recent study reported increases in neurotransmitter levels in the frontal cortex following ultrasound stimulation of the rat thalamus, a key relay station for sensory and motor information [3]. These animal studies support the idea that TUS can affect brain physiology in a region-specific manner, and is even able to reach deep areas of the brain far from the perimeter of the skull. It didn’t take long for the incentive of novel therapeutic use to fuel the debate over whether one should try aiming ultrasound at a human’s brain.
Human neuromodulation studies
The first human study attempted to use TUS to assess its effects on self-reported pain and mood in a geriatric population of chronic pain patients [4]. The experiment utilized a medical-grade ultrasound imaging device routinely used for other procedures. In addition, the device had the ability to output pulses at an intensity well-within FDA guidelines, posing minimal risk for the patients in regards to any long-term side-effects. Researchers aimed the device over the temporal region of the skull, on the opposite side from the source of reported body pain, and delivered a single fifteen second dose of TUS. While self-reported pain was not altered by TUS, the patients who received TUS reported an improvement in mood compared to placebo controls at both 10 and 40 minutes after the stimulation session. These results opened the floodgates to even more questions. Why did TUS have an ameliorating effect on mood and not pain? Was this therapy only suitable for older, chronic-pain patients? What about those who are healthy? Despite the study’s limitations, the brave souls who participated in this study set the stage for preliminary data suggesting that TUS could be developed as a treatment for mood disorders. Interestingly, a follow-up study replicated this mood-improvement effect on a college-student population [5] . Overall, these two studies invite the possibility of TUS stimulation as a therapeutic for mood-disorder patients who are resistant to conventional pharmacological treatment.

Example locations for transcranial ultrasound stimulation. Location #5 is representative of where Hameroff S et al (2013) applied stimulation pulses, always opposite of the source of a patient’s chronic pain.
The search for a mechanism
Determining the direct effects of TUS stimulation on the brain has proven to be a challenge. There is speculation that the mechanic vibrations from TUS alter the microtubules in a neuron’s cytoskeleton [6]. Microtubules maintain the structure of a cell, and their resonance frequencies happen to be in the MHz range, indicating a susceptibility to TUS mechanical forces. It may be possible that the mechanical effects of TUS on neuron microtubules change the cell’s propensity to become active and fire. However, if TUS is to be touted as a therapeutic tool, then greater targeting specificity must be achieved, largely due to the heterogeneity in neuronal populations implicated in brain-related diseases. While the precise mechanism for TUS remain poorly understood, other avenues for investigating its effects on the central nervous system involve an unlikely animal model: the nematode (Caenorhabditis elegans).

A lot goes on in C. elegans. Their mapped out nervous system provides a powerful tool for genetic manipulation.
Researchers at the Salk Institute are developing an ultrasound stimulation method to target individual neurons. Using “sonogenetics”, they identified a pore-forming subunit of a mechanotransduction channel, TRP-4, that was shown to be responsive to low-pressure ultrasound [7]. C. elegans reversed their direction of movement after a single pulse of ultrasound stimulation. However, this effect required the presence of microbubbles in the testing plate, which are believed to be amplifying the ultrasound signals to activate TRP-4. Unfortunately, mammalian brains have no homologues of TRP-4 channel, yet current research in this lab has begun to consider genetically modifying mice to carry a mutated TRP-4 that could potentially be activated by ultrasound stimulation. If microbubbles are a necessity in mammals as in C. elegans, they can be safely administered intravenously to circulate a mammal’s body up to the brain. If their method works, it’ll be the beginning of establishing a non-invasive method that selectively activates genetically modified neurons, ramping up the specificity of stimulation without the need of intracranial implants. However, this would also require you to have genetically modified neurons! I’ll have a huge amount of respect to those who volunteer to participate in these future studies.

Set up for the experiments conducted in Ibsen S et al (2015). On the left is the behavioral effect of ultrasound stimulation in the presence of microbubbles.
While ultrasound may conjure the image of bat echolocation or pregnancy imaging procedures, TUS is hopefully not too far off from its use as a neurological therapeutic. Over the next decade, it’ll be interesting to keep track of how animal studies pinpoint the direct, biological effect of ultrasound on neurons. Until then, as the aforementioned human studies have shown, it’s worth to keep assessing the method’s mood-alleviating outcomes. At some point, both research directions will converge, and the optimal brain stimulation parameters for different ailments could be worked out. Someday, a visit to the doctor may end up with a prescription for ultrasound brain stimulation!
Sources:
Figure 1: Sciencedaily article
Figure 2: Slideshare
Figure 3: Hameroff S et al. Brain Stimul 2013;6(3):409–15.
Figure 4: Worm Atlas
Figure 5. Ibsen S et al. Nature communications 6, 8264
- Foley JL, Vaezy S, Crum LA. Applications of high-intensity focused ultrasound in medicine: Spotlight on neurological applications. Appl Acoust 2007;68(3):245–59
- Tyler WJ, Tufail Y, Finsterwald M, Tauchmann ML, Olson EJ, Majestic C. Remote excitation of neuronal circuits using low intensity, low-frequency ultrasound. PLoS One 2008;3(10).
- Min B-K, Yang PS, Bohlke M, Park S, R.Vago D, Maher TJ et al. Focused ultrasound modulates the level of cortical neurotransmitters: Potential as a new functional brain mapping technique. Int J Imaging Syst Technol 2011;21(2):232–40
- Hameroff S, Trakas M, Duffeld C, Annabi E, Gerace MB, Boyle P et al. Transcranial ultrasound (TUS) effects on mental states: a pilot study. Brain Stimul 2013;6(3):409–15
- Sanguinetti JL, Smith EE, Dieckman L, Vanuk J, Hameroff S, Allen JJB. Transcranial ultrasound for brain stimulation: Effects on mood. Psychophysiology 2013;50:S46
- Sanguinetti, Joseph L. et al. “Human Brain Stimulation with Transcranial Ultrasound Potential Applications for Mental Health.” (2014).
- Ibsen, S., Tong, A., Schutt, C., Esener, S. & Chalasani, S. H.Sonogenetics is a non-invasive approach to activating neurons in Caenorhabditis elegans. Nature communications 6, 8264, doi: 10.1038/ncomms9264 (2015).

Pingback: Feelin’ the Beat (Frequency) | NeuWrite San Diego