 June
20
June
20
Tags
Exosomes: Helping you stay in touch with your trillions of closest friends.
There are roughly 37.2 trillion cells in the human body. That’s nearly 5 thousand times the total number of people on the entire planet, and all these cells must somehow work together to make you “You.” Each cell has a set role to play in keeping you healthy and functioning, but how do these cells know what their role is? How do they communicate with the other trillions of cells to keep everything flowing correctly? These questions have many long and complicated answers. There are many types of signals in the body. For instance, there are well regulated signals, called hormones, which are circulated throughout the bloodstream as well as electrical signals used by neurons to communicate with each other and with muscles.
Over the past thirty years or so, researchers have discovered and begun investigating a new type of signal: a way cells of any kind can send out little messages to the rest of the body about who they are, how they are doing, and generally what is going on in their internal world. These little message bundles are called exosomes, and they are essentially like the Facebook of the body. These tiny membrane bound vesicles are released by the cell and contain RNA and proteins that are specific to the cell releasing them. The cargoes of these exosomes are not only reflective of the type of cell releasing them but also the current state of that cell, as the protein and RNA composition of a cell vary depending on what is happening in that cell. These little vesicles get released into the surrounding space of the cell and end up in the fluids of the body like the blood, urine, and saliva allowing them to be transported anywhere and everywhere. It is like you taking a picture of some event going on in your life at a particular time and then posting it to social media where it could end up anywhere as a snapshot of that moment in your life. Since exosomes are transported via bodily fluids, these little snapshots can then end up throughout the entire body. Just like that embarrassing picture you know will always exist out in the abyss of the internet.
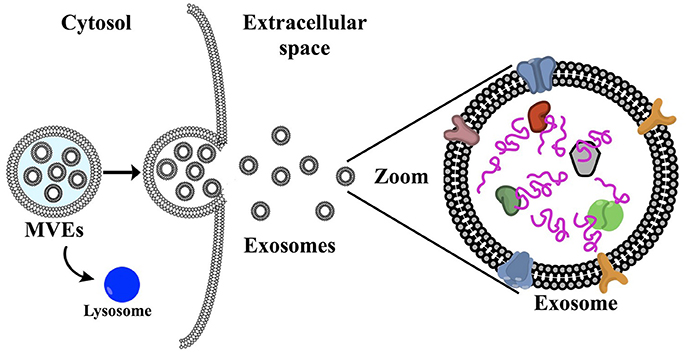
The study of exosomes has exploded over the past three decades. Originally they were thought to be simply a means for cells to get rid of waste, but now we know that most certainly is not the case! Vast scores of research has come out showing some of the complex, intricate roles played by exosomes in cell-to-cell communication in health and disease that we are only just beginning to understand. The early and more traditional context in which exosomes were studied was in cancer biology, but the field has expanded to a vast range of tissues and diseases. I will focus here on a few of their roles and implications in the nervous system specifically. These include exosome transfer of tau protein in Alzheimer’s disease, using exosomes to diagnose disease, as well as the use of exosomes to treat disease.
The role of exosomes in Alzheimer’s disease
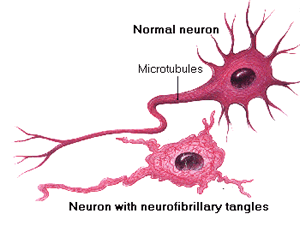
Alzheimer’s disease is incredibly complex and scientists still don’t fully understand how the brain damage progresses or even where it begins. Both of these topics are still up for debate. We do know, however, that aggregates containing the protein tau are one of the major hallmarks of Alzheimer’s disease. Misfolded tau protein builds up in the neurons of Alzheimer’s patients’ brains, causing cell death. This misfolded tau protein then propagates and spreads to other neurons, eventually affecting more and more of the brain. While these tau aggregates may spread in a number of different ways, one way they may do so is via exosome secretion and uptake by neighboring cells. Work by Asai, et al, 2015 as well as by Polanco et al, 2016 has demonstrated that depletion of exosome formation can halt the spread of tau and that exosomes containing tau can cause new tau aggregate formation in new cells.
Exosomes as a non-invasive diagnostic tool
Exosomes are little bundles of stuff reflective of their cell of origin. When the cell undergoes changes, say in the case of a disease state, the composition of the exosomal cargo changes as well. This has incredible clinical implications, as exosomes may be able to serve as early biomarkers for clinical pathologies. By obtaining small volumes of bodily fluid (such as blood, urine or cerebral spinal fluid), the cargoes of the exosomes in that fluid can be analyzed for specific changes indicative of a disorder. This will allow for very non-invasive diagnosis of disease. Since obtaining bodily fluids is so cheap and easy, more individuals will be able to be monitored for specific changes in their exosome cargo composition, especially if they have a family history of certain diseases. These tools are starkly different than many of the current tools used for many diagnostic practices, such as tissue biopsies. Imagine, rather than being admitted into the hospital, being put under anesthesia, having an incision made, and having doctors physically cut out a piece of tissue, instead just going into a clinic and having some blood drawn or peeing in a cup. This drastic rise in availability will likely mean earlier disease detection, which has the potential to lead to drastic improvements in treatment success.
One such instance where exosomes have been found to be potentially useful in the diagnosis of disease is in the very progressive brain cancer, glioblastoma. In 2008, Skog and coworkers detected a glioblastoma-specific protein called epidermal growth factor receptor VIII (EGFRVIII) in exosomes taken from the serum of patients. This indicates that exosomal EGFRVIII may provide early diagnostic information for glioblastoma. Rather than waiting for a tumor to get large enough to be detected by magnetic resonance imaging (MRI) and then getting a biopsy of the brain tissue, exosomes may offer the potential for patients to be diagnosed from a very early stage simply with a small volume of blood. Currently adults with the more aggressive glioblastoma, given traditional treatment, have a median survival of about 14.6 months, making it one of the most lethal among cancers. With accelerated and improved diagnostic tools, like exosome profiling, patients may be able to start treatment sooner and live longer, healthier lives.
Big treatments can come in small packages
Exosomes transport cell type-specific cargo over large distances. When these exosomes are taken up by other cells, these cargoes can elicit different effects depending on the type and condition of the cell taking them in. Because exosomes, carrying specific cargo, can have such drastic effects on other cell types, they offer great hope for the development of therapies. If clinicians specifically block or enhance exosomal interactions between cells, depending on what those exosomes do, this could have profound effects on disease progression and outcomes. In the case of stroke, exosomes released by compromised neurons seem to play a role in the remodeling of the nervous system. More generally, exosomes contribute to local changes in neuron wiring and also influence neuronal networks by long-range communication within the central nervous system. In stroke patients it seems damaged cells release exosomes that have a negative effect on surrounding brain tissue. Chen and Chopp, 2018 showed the inhibition of exosomal release from damaged cells may prove to have a therapeutic effect on patients. There is also growing evidence that exosomes with certain cargo may themselves serve in regeneration and repair and, therefore, may be exploited in treatments for a huge range of diseases.
Perhaps a not-so-distant future
I imagine a world one day where routine blood draws will include exosome profiling to look for a whole array of disease markers. This will allow doctors to detect changes that have not even begun to be noticed by patients or even detectable by other diagnostic tools. In this world exosomes will be created with specific cargo and infused into the bloodstream of patients to help treat an assortment of diseases such as chronic pain, and various neurodegenerative disorders, just to name a few. While this may seem too good to be true, and maybe even to some like a chapter in a futuristic sci-fi novel, we are getting closer than ever to making this dream a reality.
References
Abta.org. (2019). Available at: https://www.abta.org/wp-content/uploads/2018/03/glioblastoma-brochure.pdf [Accessed 14 Jun. 2019].
Asai, H., Ikezu, S., Tsunoda, S., Medalla, M., Luebke, J., Haydar, T., Wolozin, B., Butovsky, O., Kügler, S. and Ikezu, T. (2015). Depletion of microglia and inhibition of exosome synthesis halt tau propagation. Nature Neuroscience, 18(11), pp.1584-1593.
Chen, J. and Chopp, M. (2018). Exosome Therapy for Stroke. Stroke, 49(5), pp.1083-1090.
de la Torre Gomez, C., Goreham, R., Bech Serra, J., Nann, T. and Kussmann, M. (2018). “Exosomics”—A Review of Biophysics, Biology and Biochemistry of Exosomes With a Focus on Human Breast Milk. Frontiers in Genetics, 9.
Ebrahimkhani, S., Vafaee, F., Hallal, S., Wei, H., Lee, M., Young, P., Satgunaseelan, L., Shivalingam, B., Suter, C., Buckland, M. and Kaufman, K. (2018). Deep sequencing of circulating exosomal microRNA allows non-invasive glioblastoma diagnosis.
Eveleth, R. (2019). There are 37.2 Trillion Cells in Your Body. [online] Smithsonian. Available at: https://www.smithsonianmag.com/smart-news/there-are-372-trillion-cells-in-your-body-4941473/ [Accessed 14 Jun. 2019].
Harding, C., Heuser, J. and Stahl, P. (2013). Exosomes: Looking back three decades and into the future. The Journal of Cell Biology, 200(4), pp.367-371.
Kirchgessner, M. (2019). Glioblastoma: John McCain’s final battle. [online] NeuWrite San Diego. Available at: https://neuwritesd.org/2018/09/20/glioblastoma-john-mccains-final-battle/ [Accessed 14 Jun. 2019].
Lilt.ics.hawaii.edu. (2019). Aluminum Role in NFT Formation. [online] Available at: http://lilt.ics.hawaii.edu/collabreps/materials/main/articles/AlCausesNFTsInRats.php [Accessed 20 Jun. 2019].
Mandelkow, E. (1998). Tau in Alzheimer’s disease. Trends in Cell Biology, 8(11), pp.425-427.
Polanco, J., Scicluna, B., Hill, A. and Götz, J. (2016). Extracellular Vesicles Isolated from the Brains of rTg4510 Mice Seed Tau Protein Aggregation in a Threshold-dependent Manner. Journal of Biological Chemistry, 291(24), pp.12445-12466.
Pooler, A., Polydoro, M., Wegmann, S., Nicholls, S., Spires-Jones, T. and Hyman, B. (2013). Propagation of tau pathology in Alzheimer’s disease: identification of novel therapeutic targets. Alzheimer’s Research & Therapy, 5(5), p.49.
Shafiei, S., Guerrero-Muñoz, M. and Castillo-Carranza, D. (2017). Tau Oligomers: Cytotoxicity, Propagation, and Mitochondrial Damage. Frontiers in Aging Neuroscience, 9.
Skog, J., Würdinger, T., van Rijn, S., Meijer, D., Gainche, L., Curry, W., Carter, B., Krichevsky, A. and Breakefield, X. (2008). Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nature Cell Biology, 10(12), pp.1470-1476.
Solders, S. (2019). Where does Alzheimer’s disease begin?. [online] NeuWrite San Diego. Available at: https://neuwritesd.org/2018/07/05/where-does-alzheimers-disease-begin/ [Accessed 14 Jun. 2019].
Spencer, B. (2019). From symptoms to biology: shifting definitions of Alzheimer’s disease. [online] NeuWrite San Diego. Available at: https://neuwritesd.org/2018/11/15/from-symptoms-to-biology-shifting-definitions-of-alzheimers-disease/ [Accessed 14 Jun. 2019].
Wang, J., Sun, X., Zhao, J., Yang, Y., Cai, X., Xu, J. and Cao, P. (2017). Exosomes: A Novel Strategy for Treatment and Prevention of Diseases. Frontiers in Pharmacology, 8.
Xiao, T., Zhang, W., Jiao, B., Pan, C., Liu, X. and Shen, L. (2017). The role of exosomes in the pathogenesis of Alzheimer’ disease. Translational Neurodegeneration, 6(1).
Yalcinbas, E. (2019). Battling A Brain Tumor The Loki Way. [online] NeuWrite San Diego. Available at: https://neuwritesd.org/2018/03/03/battling-a-brain-tumor-the-loki-way/ [Accessed 14 Jun. 2019].
Zhang, Z., Buller, B. and Chopp, M. (2019). Exosomes — beyond stem cells for restorative therapy in stroke and neurological injury. Nature Reviews Neurology, 15(4), pp.193-203.

You must be logged in to post a comment.